J.ophthalmol.(Ukraine).2022;6:24-29.
|
http://doi.org/10.31288/oftalmolzh202262429 Received: 28.08.2022; Accepted: 31.10.2022; Published on-line: 21.12.2022 Ophthalmological involement and laboratory changes in patients with ANCA-associated vasculitis and polyarteritis nodosa O. Iaremenko, L. Petelytska Bogomolets National Medical University; Kyiv (Ukraine) TO CITE THIS ARTICLE: Iaremenko O, Petelytska L. Ophthalmological involvement and laboratory changes in patients with anca-associated vasculitis and polyarteritis nodosa. J.ophthalmol.(Ukraine).2022;6:24-29. http://doi.org/10.31288/oftalmolzh202262429
Background. Ophthalmic involvement (OI) is an early manifestation of systemic vasculitis and may lead to blindness. Purpose. Aim of the study was to identify the frequencies and patterns of OI in patients with ANCA-associated vasculitis (AAV) and polyarteritis nodosa (PAN) and evaluate the laboratory changes depending on OI. Material and Methods. This work included retrospective study (112 patients with PAN and 80 patients with AAV) and prospective cohort study, which evaluated serum levels of endothelin-1 (ET-1) in 36 patients and 26 healthy сontrols. Results. OI at diagnosis in AAV occurred 4 times more often compared to PAN and consisted of conjunctivitis/episcleritis (16% AAV patients), optic neuropathy (5% PAN patients), orbital mass, uveitis, scleritis, epiphora or dry eyes. OI was more commonly detected in PR3-ANCA(+) vs PR3-ANCA(-) patients. The levels of ET-1 were elevated in patients without OI compared with control group and patients with OI, but ability of ET-1 to differentiate such patients was poor. Conclusion. The most common OI in patients with AAV was episcleritis. Optic neuropathy was the only manifestation of OI in patients with PAN. OI were more commonly detected in PR3-ANCA(+) vs PR3-ANCA(-) patients. The serum levels of ET-1 were significantly elevated in patients without OI compared with patients with OI and control group, but its diagnostic value was not approved in the ROC analysis. Keywords: eye involvement, anti-neutrophil cytoplasmic antibody-associated vasculitis, granulomatosis with polyangiitis, eosinophilic granulomatosis with polyangiitis, microscopic polyangiitis, polyarteritis nodosa Introduction. ANCA-associated vasculitis (AAV) and polyarteritis nodosa (PAN) are systemic vasculitis with inflammation and necrosis of small and medium vessels in multiple organs. [1]. The clinical manifestations vary from local form with single-organ involvement to multisystem life-threatening patterns. The two types of ANCA are generally distinguished, antibodies against proteinase-3 (PR3), which are typical for granulomatosis with polyangiitis (GPA), and antibodies against myeloperoxidase (MPO), which are predominantly detected in patients with microscopic polyangiitis (MPA) and some patients with eosinophilic granulomatosis with polyangiitis (EGPA). [2]. Negative PR3-ANCA and MPO-ANCA do not exclude the diagnosis of ANCA-associated vasculitis. The limited form of vasculitis is often ANCA-negative. Different clinical manifestations of vasculitis are associated with a certain frequency of detection of ANCA. According to Lionaki S. et al., the majority of patients with isolated renal disease were positive for MPO-ANCA (81%), while almost all patients with bone destruction or saddle-shaped deformity of the nose showed PR3-ANCA (94%). [3]. Ophthalmological manifestations, including ocular or orbital involvement, are common clinical signs of systemic vasculitis that occur in 16-52% of patients with AAV and in 10–20% of patients with PAN. [4-6]. Eye involvement is an initial presenting finding in 8% to 16% of patients. [6-8]. Ocular involvement is frequently seen in GPA, but rarely in EGPA and MPA. [9]. Gender predominance of ophthalmological manifestation is still discussed. Some studies have reported a male predominance of 57-63% [4, 10], the others have shown a female predominance of 51 -72%. [5, 11]. All parts of the eye and orbit can be affected; however, scleritis and orbital mass are the most common presenting manifestation. Conjunctivitis, peripheral ulcerative keratitis, uveitis, choroidal disease, and retinal vasculitis can be also the clinical features of systemic vasculitis. The orbital mass formation is a clinical indication for ANCA testing. [12]. Eye damage can lead to blindness and significant socio-economic consequences. The mortality rate of patients with ocular manifestations in AAV is 2.75-fold higher than patients with other inflammatory eye disorders [10]. That’s why the early recognitions of ocular disease and evaluation of other signs of systemic process are the corner stone in diagnosis of AAV and PAN. Most of the previous studies of ophthalmological manifestations of AAV were limited to case reports, case series or retrospective studies. There are no specific laboratory markers for ocular involvement in systemic vasculitis. Endothelin-1 (ET-1) is a potential participant in the local regulation of intraocular pressure, ocular blood vessel tone, and iris smooth muscle tone, suggesting that it may be an important mediator in the development of ocular pathologic conditions. [13]. Lower ET-1 plasma levels were found in the optic neuropathy. [14]. The objectives of the present observational study in cohort of patients with AAV and PAN were to analyze the frequencies and patterns of ophthalmic involvement, and laboratory characteristics. Material and methods Data source and study population This work included retrospective and prospective studies, which were conducted at a rheumatology department of two Kyiv tertiary hospitals. Retrospective study included identification of medical records of potential patients with AAV and PAN treated from 1979 to 2013 from the clinical database. Selected medical records were reviewed to confirm the diagnosis of AAV or PAN according to the ACR 1990 criteria [15, 16, 17] and definition of the Chapel Hill Consensus Conference 2012. [1]. The search process is shown in Fig.1. Confirmed patient’s data on demographics (sex and age at diagnosis), initial presentation signs, systemic and ophthalmological manifestations and laboratory features (ESR, CRP, eosinophilia, serum creatinine level and ANCA, including PR3-ANCA and MPO-ANCA). Patients with ANCA-associated vasculitis usually were analyzed. Clinical activities of patients were calculated according to the Birmingham Vasculitis Activity Score (BVAS), version 3. [18].
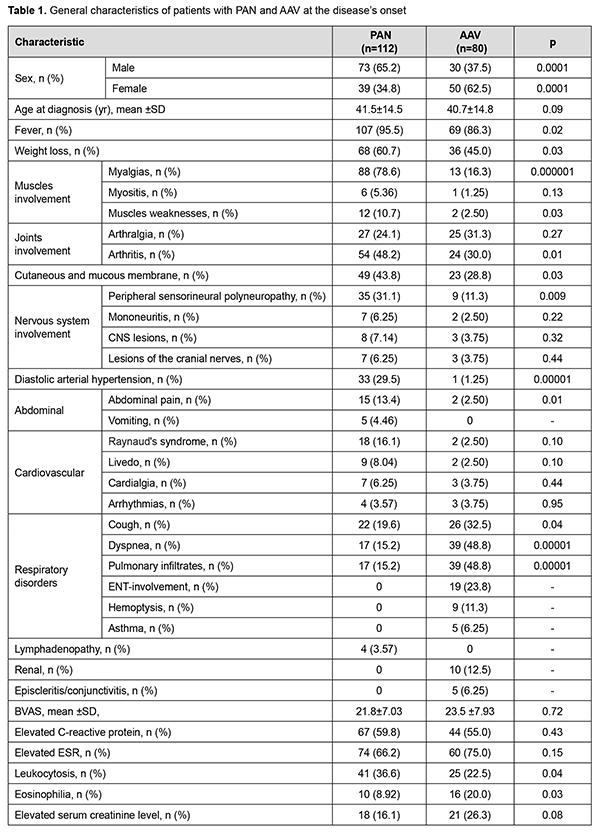
Prospective study included 36 patients with systemic vasculitis (28 patients with AAV and 8 patients with PAN) with and without eye involvement and 26 healthy controls seen from 2013 to 2018. Ophthalmological manifestations were present in 17 patients. Persons of healthy control did not have any autoimmune or inflammatory disease or cardiovascular disease, or take immune-suppressive treatment. The serum levels of endothelin-1 (ET-1), pmol/L, were determined by immunoassay analysis using the kits of Biomedica (USA). The outcomes of this study were the differences in marker levels between patients with and without eye involvement and healthy controls. All the patients with ophthalmologic involvement had been seen at least once by an ophthalmologist. Approval for this study was obtained from the Bogomolets National medical university review board (approval number 28/20). According to the conclusion of the bioethical commission, the study did not contain an increased risk for the subjects of the study and was performed taking into account the existing bioethical norms and scientific standards regarding the conduct of clinical research involving patients. All procedures in this study were performed in accordance with 1964 Helsinki declaration and its later amendments. Statistical analysis Data are presented as mean (standard deviation) for continuous variables with normal distribution and as median (range) for continuous variables with abnormal distribution or number (%) for qualitative variables. Distributions of marker values were evaluated for normality using Shapiro–Wilk test. Comparisons were conducted using the Chi-square test or the Fisher’s exact test for categorical variables and Student’s t-tests for continuous variables. Differences in laboratory marker levels between patients and healthy controls were estimated by analysis of the absolute changes in marker levels. Receiver operating characteristic (ROC) analysis was used for evaluation of diagnostic significance of laboratory data. The area under ROC curve (AUC) and 95% confidence intervals for ET-1was calculated. P <0.05 was considered statistically significant. Analyses were performed using SPSS software, v.15.0.1. Results The most frequent clinical manifestations at PAN onset were fever (96%), myalgia (79%), weight loss (61%), arthritis (48%), skin lesions (44%) and peripheral polyneuropathy (31%). Asthma, upper respiratory tract involvement, hemoptysis and glomerulonephritis were reported only in patients with AAV. There were no significant differences between patients with PAN and AAV in the median age of onset and clinical activity according to BVAS (Table 1).
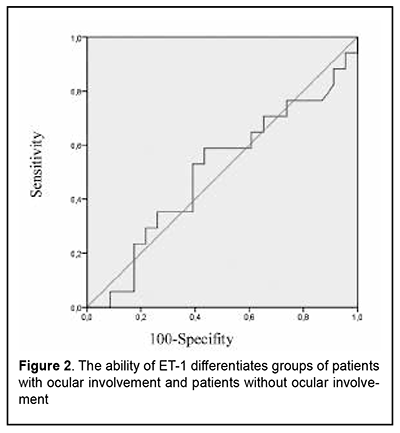
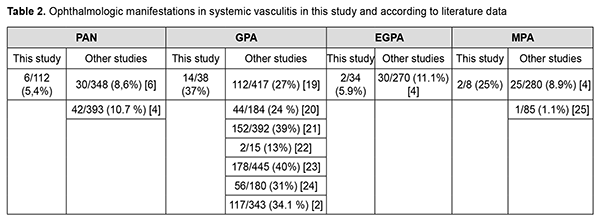
Ophthalmological manifestation (conjunctivitis/episcleritis) at the disease’s onset was present in 3 patients with GPA and one patient with EGPA and MPA. Two patients with PAN had optic neuropathy as an initial sign. Ocular symptoms were combined with the others systemic features in all patients. Ophthalmological involvement at diagnosis consisted of conjunctivitis/episcleritis (10 GPA, 2 MPA, and 1 EGPA patients), optic neuropathy (6 PAN patients), orbital mass (2 GPA patients), uveitis (1 GPA patient), scleritis (1 GPA patient), epiphora (1 GPA patient) or dry eyes (1 EGPA patient). Most often eye involvement occured in patients with GPA (in 37% of patients). Gender and age differences in the incidence of ophthalmic manifestations of GPA were not detected. In general, at the time of diagnosis, ocular manifestations in AAV occurred 4 times more often compared to PAN (22.5% vs 5.4%, p=0.0004). Optic neuropathy was the only manifestation of eye lesions in patients with PAN. Ocular changes were more commonly detected in PR3-ANCA positive GPA patients (50%) in comparison with PR3-ANCA negative GPA patients (15%, p <0.05). PR3-ANCA positive GPA patients did not differ significantly from PR3-ANCA negative GPA patients in the BVAS activity index (24.8 vs 20.46, respectively) and demographic characteristics (age - 47.1 vs. 41.3 years, men, % - 33.3 vs. 38.5, respectively). The ocular manifestations of 36 patients with systemic vasculitis included in prospective study were presented by episcleritis (n=10), anterior uveitis (n=3), ischemic optic neuritis (n=3, in GPA patients) and occlusive retinal vasculitis (n=1). Eye involvement was an initial presentation in 14% of patients. All patients had active disease (BVAS>11). Patients with and without eye involvement did not differ significantly according to BVAS, ANCA, ESR and CRP. The level of ET-1 (mean ±SD) in patients with eye involvement (n=17) was 0.28 ± 0.13. There was no significant difference in ET-1 level between patients with eye involvement and control group (0.27 ± 0.10, p> 0.05). At the same time, in patients without eye involvement (n=19), it was significantly elevated (0.36 ± 0.34) compared with control group and with group of patients with eye involvement (p<0.001). ROC analysis (Fig.2) showed that the AUC for ET-1 is 0.5±0.10 (95% CI 0.32-0.69, p=0.98), which indicates not acceptable capacity for ET-1 to differentiate groups of patients with ocular involvement and patients without ocular involvement (sensitivity - 59%, specificity - 57%).
Discussion Ocular involvement in systemic vasculitis ranges from mild to blindness- threatening without appropriate treatment. The "red eye" sign may occur in variable ophthalmological manifestations (conjunctivitis, blepharitis, episcleritis, scleritis, keratitis) with different prognoses. That’s why patients with suspected systemic vasculitis should be referred to an ophthalmologist. It should be kept in mind that ocular manifestations are not specific only for AAV or PAN and should be differentiated from other vasculitis, inflammatory and infectious processes. The results of this retrospective study of systemic vasculitis patients from rheumatology departments presented the frequencies of different ocular and orbital involvements at disease onset and diagnosis of AAV or PAN. Generally, the ocular signs were detected in 6.3% of patients with AAV and 1.8 % of patients with PAN at disease onset and in 22.5% AAV and 5.4 % PAN at diagnosis (Table 2). Most often eye involvement occurs in patients with GPA (in 37% patients) and corresponds to the literature data (ranging from 13 to 40%). [19-23]. Gender and age differences in the incidence of ophthalmic manifestations in the onset of GPA were not detected. Literature data about gender differences in the incidence of ophthalmic manifestations of GPA is still controversial. [4, 5, 8, 10].
The most common type of ocular manifestation in this cohort was episcleritis, particularly in PR3-ANCA-positive patients, which is in line with previous studies. [5, 7]. The only ophthalmic manifestation of PAN was optic neuropathy, which was detected in 5% of patients and represented by anterior ischemic optic neuropathies. Inflammatory optic neuritis was present also in few of our GPA patients in prospective study. The frequency of orbital mass or pseudotumour was not high in our cohort and occurred only in two patients. The biopsy for conformation of granuloma or vasculitis was not performed. Due to literature data, 30% of patients with orbital mass did not have other organ involvement at the time of AAV diagnosis and one-third of these patients had negative ANCA. [5, 26]. The signs of epiphora or dry eyes can develop in patients with AAV [27]. These manifestations were present in patients in our study. The epiphora appears due to involvement of the lacrimal sac secondary to inflammatory prosses of the surrounding sinuses. Dry eyes may be provoked by granulomatosis inflammation of the lacrimal glands. The serum levels of ET-1 were lower in AAV and PAN patients with eye involvement compared with patients without eye involvement, but this cannot be used for diagnostic purposes. Our study has some strengths and limitations. We describe here the unique study of ophthalmological manifestation of AAV and PAN in Ukraine. Limitations include retrospective design of the first study that may have influence on completeness of data and a relatively small number of patients including in prospective analysis that requires combination of all AAV and PAN in one group. Ophthalmological examinations were performed only for patients with ocular complaints. That’s why our study might have underestimated the frequencies and missed patients with isolated eye involvement, which were treated only by ophthalmologist. Follow-up ophthalmological examinations were not performed. In conclusion, the most common ocular manifestation in patients with AAV was episcleritis, which occurred in almost one third of GPA patients. However, scleritis, uveitis, optic neuropathies and orbital mass were registered in systemic vasculitis. Ophthalmic findings in MPA, EGPA, and PAN are much rarer. Optic neuropathy is the only manifestation of eye lesions in patients with PAN. Ocular disease generally occurred in PR3-ANCA positive patients with other systemic manifestations. The serum level of ET-1 was significantly elevated in patients without eye involvement compared with patients with eye involvement and healthy controls, but it didn’t have significant diagnostic value according to the ROC analysis. The management of AAV and PAN patients with ophthalmological manifestations required combined efforts of a rheumatologist with an ophthalmologist. Acknowledgements These data were presented as a poster presentation and abstract at Annual European Congress of Rheumatology, Madrid, Spain (12–15 June 2019) [28].
References 1.Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis and rheumatism. 2013; 65(1): 1–11. 2.Millet A, Pederzoli-Ribeil M, Guillevin L, Witko-Sarsat V, Mouthon L. Antineutrophil cytoplasmic antibody-associated vasculitides: is it time to split up the group? Annals of the rheumatic diseases. 2013; 72(8): 1273–1279. 3.Lionaki S, Blyth ER, Hogan SL, Hu Y, Senior BA, Jennette CE et al. Classification of antineutrophil cytoplasmic autoantibody vasculitides: the role of antineutrophil cytoplasmic autoantibody specificity for myeloperoxidase or proteinase 3 in disease recognition and prognosis. Arthritis and rheumatism. 2012; 64(10): 3452–3462. 4.Rothschild P R, Pagnoux C, Seror R, Brézin AP, Delair E, Guillevin L. Ophthalmologic manifestations of systemic necrotizing vasculitides at diagnosis: a retrospective study of 1286 patients and review of the literature. Seminars in arthritis and rheumatism. 2013; 42(5): 507–514. 5.Ungprasert P, Crowson CS, Cartin-Ceba R, Garrity JA, Smith WM, Specks U et al. Clinical characteristics of inflammatory ocular disease in anti-neutrophil cytoplasmic antibody associated vasculitis: a retrospective cohort study. Rheumatology (Oxford, England). 2017; 56(10) : 1763–1770. 6.Pagnoux C, Seror R, Henegar C, Mahr A, Cohen P, Le Guern V et al. Clinical features and outcomes in 348 patients with polyarteritis nodosa: a systematic retrospective study of patients diagnosed between 1963 and 2005 and entered into the French Vasculitis Study Group Database. Arthritis and rheumatism. 2010; 62(2): 616–626. 7.Pakrou N, Selva D, Leibovitch I. Wegener's granulomatosis: ophthalmic manifestations and management. Seminars in arthritis and rheumatism. 2006; 35(5): 284–292. 8.Hoffman GS, Kerr GS, Leavitt RY, Hallahan CW, Lebovics RS, Travis WD et al. Wegener granulomatosis: an analysis of 158 patients. Annals of internal medicine. 1992; 116(6): 488–498. 9.Kubal AA, Perez VL. Ocular Manifestations of ANCA-associated Vasculitis. Rheumatic Disease Clinics of North America. 2010; 36(3): 573–586.Crossref PubMed 10.Watkins AS, Kempen JH, Choi D, Liesegang TL, Pujari SS, Newcomb C et al. Ocular disease in patients with ANCA-positive vasculitis. Journal of ocular biology, diseases, and informatics. 2009; 3(1): 12–19. 11.Miyanaga M, Takase H, Ohno-Matsui K. Anti-Neutrophil Cytoplasmic Antibody-Associated Ocular Manifestations in Japan: A Review of 18 Patients. Ocular immunology and inflammation. 2021; 29(5): 991–996. 12.Savige J, Davies D, Falk RJ, Jennette JC, Wiik A. Antineutrophil cytoplasmic antibodies and associated diseases: a review of the clinical and laboratory features. Kidney international. 2000; 57(3): 846–862. 13.Sugiyama T. Involvement of Endothelin-1 in the Pathophysiology of Normal-Tension Glaucoma. International Journal of Ophthalmic Research. 2015; 1(2): 36-40. 14.Ohguro H, Mashima Y, Nakazawa M. Low levels of plasma endothelin-1 in patients with retinitis pigmentosa. Clinical ophthalmology (Auckland, N.Z.). 2010; 4: 569–573. 15.Lightfoot RW Jr, Michel BA, Bloch DA, Hunder GG, Zvaifler NJ, McShane DJ et al. The American College of Rheumatology 1990 criteria for the classification of polyarteritis nodosa. Arthritis and rheumatism. 1990; 33(8): 1088–1093. 16.Leavitt RY, Fauci AS, Bloch DA, Michel BA, Hunder GG, Arend WP et al. The American College of Rheumatology 1990 criteria for the classification of Wegener's granulomatosis. Arthritis and rheumatism. 1990; 33(8): 1101–1107. 17.Masi AT, Hunder GG, Lie JT, Michel BA, Bloch DA, Arend WP et al. The American College of Rheumatology 1990 criteria for the classification of Churg-Strauss syndrome (allergic granulomatosis and angiitis). Arthritis and rheumatism. 1990; 33(8): 1094–1100. 18.Mukhtyar C, Lee R, Brown D, Carruthers D, Dasgupta B, Dubey S et al. Modification and validation of the Birmingham Vasculitis Activity Score (version 3). Annals of the rheumatic diseases. 2009; 68(12): 1827–1832. 19.Wójcik K, Wawrzycka-Adamczyk K, Włudarczyk A, Sznajd J, Zdrojewski Z, Masiak A. et al. Clinical characteristics of Polish patients with ANCA-associated vasculitides-retrospective analysis of POLVAS registry. Clinical rheumatology. 2019 ; 38(9): 2553–2563. 20.Solans-Laqué R, Fraile G, Rodriguez-Carballeira M, Caminal L, Castillo MJ, Martínez-Valle F et al. Spanish Registry of systemic vasculitis (REVAS) from the Autoimmune Diseases Study Group (GEAS) of the Spanish Society of Internal Medicine (SEMI) (2017). Clinical characteristics and outcome of Spanish patients with ANCA-associated vasculitides: Impact of the vasculitis type, ANCA specificity, and treatment on mortality and morbidity. Medicine. 2017; 96(8): e6083. 21.Mahr A, Katsahian S, Varet H, Guillevin L, Hagen EC, Höglund P. et al. Revisiting the classification of clinical phenotypes of anti-neutrophil cytoplasmic antibody-associated vasculitis: a cluster analysis. Annals of the rheumatic diseases. 2013; 72(6): 1003–1010. 22.Martinez-Morillo М, Grados D. Granulomatosis With Polyangiitis (Wegener). Description of 15 Cases. Reumatol Clin. 2012; 8: 15–19. 23.Holle JU, Gross WL, Latza U, Nölle B, Ambrosch P, Heller M. et al. Improved outcome in 445 patients with Wegener's granulomatosis in a German vasculitis center over four decades. Arthritis and rheumatism. 2011; 63(1): 257–266. 24.Stone JH, Wegener's Granulomatosis Etanercept Trial Research Group. Limited versus severe Wegener's granulomatosis: baseline data on patients in the Wegener's granulomatosis etanercept trial. Arthritis and rheumatism. 2003; 48(8): 2299–2309. 25.Guillevin L, Durand-Gasselin B, Cevallos R, Gayraud M, Lhote F, Callard P et al. Microscopic polyangiitis: clinical and laboratory findings in eighty-five patients. Arthritis and rheumatism. 1999; 42(3): 421–430. 26.Durel CA, Hot A, Trefond L, Aumaitre O, Pugnet G, Samson M et al. Orbital mass in ANCA-associated vasculitides: Data on clinical, biological, radiological and histological presentation, therapeutic management, and outcome from 59 patients. Rheumatology. 2019; 58(9): 1565–1573. 27.Schmidt J, Pulido JS, Matteson EL. Ocular manifestations of systemic disease: Antineutrophil cytoplasmic antibody-associated vasculitis. Current Opinion in Ophthalmology. 2011; 22(6): 489–495. 28.Petelytska L, Iaremenko O, Iaremenko K. FRI0288 Ophthalmological manifestations and endothelin-1 plasma levels in patients with systemic necrotizing vasculitis (SNV)Annals of the Rheumatic Diseases. 2019;78:824-825.
Information about authors and disclosure of information Corresponding author: Liubov Petelytska – l.petelytska@gmail.com. Authors’ contributions: Oleg Iaremenko contributed to the idea for the article. Material preparation and data analysis were performed by Liubov Petelytska and Oleg Iaremenko. The first draft of the manuscript was written by Liubov Petelytska and Oleg Iaremenko commented on previous versions of the manuscript. Two authors read and approved the final manuscript. Conflict of interest: The authors have declared that no competing interests exist. Ethical approval: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Authors fulfilled the ICMJE authorship criteria. Funding information: This research does not received specific grant from any funding agency in the public, commercial or not-for -profit sector.
|