J.ophthalmol.(Ukraine).2022;6:14-18.
|
http://doi.org/10.31288/oftalmolzh202261418 Received: 02.07.2022; Accepted: 05.12.2022; Published on-line: 21.12.2022 Assessing factors of endothelial vascular dysfunction in patients with primary open-angle glaucoma A. M. Dusmukhamedova 1, D. M. Tuychibaeva 1, A. A. Khadzhimetov 2 1 Tashkent State Dentistry Institute 2 Soglonkuz Clinic Tashkent (Uzbekistan) TO CITE THIS ARTICLE: Dusmukhamedova AM, Tuychibaeva DM, Khadzhimetov AA. Assessing factors of endothelial vascular dysfunction in patients with primary open-angle glaucoma. J.ophthalmol.(Ukraine).2022;6:14-18. http://doi.org/10.31288/oftalmolzh202261418
Background: Glaucoma is a leading cause of visual disability and irreversible blindness worldwide, and significantly affects quality of life. An improvement in the early diagnosis and prevention of primary glaucoma has a special value due to increasing social significance of the disease. Purpose: To investigate the value of the factors affecting vascular endothelium (oxidative stress, complement components and blood lipid profile) in patients with primary open-angle glaucoma (POAG). Material and Methods: Ninety two patients (age, 65 to 80 years) with POAG and 30 controls (somatically healthy individuals of a similar age and no known eye disease) were included in the study. They underwent an eye examination including visual acuity, Maklakoff tonometry, non-contact tonometry, biomicroscopy, gonioscopy, and pachymetry. Plasma lipid peroxidation and antioxidant activity levels, blood lipid composition and blood C3 and C5a levels were assessed. Results: Blood malondialdehyde (MDA) levels in patients with POAG were 87% higher compared to controls. Blood C3 levels in patients with POAG were 18% higher compared to controls. The C5a complement is a multicomponent plasma enzyme system that exhibits lysis and opsonization functions during activation. Blood C5a levels were 6 times higher in patients with POAG than in controls (13.86 ± 0.44 mg/scale division versus 2.33 ± 0.11 mg/scale division). Conclusion: Increased C5a activity, hypersecretion of active oxygen species (in the presence of insufficiently efficacious antioxidant system of blood), and increased atherogenic plasma index (in the presence of low levels of high-density lipoproteins) are a mechanism of endothelial dysfunction in POAG. We found an imbalance between the prooxidant and antioxidant systems, abnormal composition of blood lipids (e.g., hypercholesterolemia and hypertriglyceridemia) and elevated C5a levels in patients with POAG. Keywords: primary open-angle glaucoma, lipid peroxidation, antioxidant system, complement components, blood lipid composition
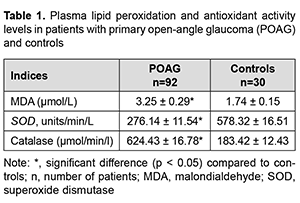
Introduction. Glaucoma is a major eye disease and a leading cause of visual disability and blindness worldwide, and has a high medical and social burden. Globally, more than 60 million people have been diagnosed as having glaucoma, with a further 105 million diagnosed as glaucoma suspects [1-3]. From 2014 through 2019, there was a 2.5-times increase in the incidence of visual disability due to glaucoma, and a 1.3 increase in the total incidence of disability in Uzbekistan [4-8]. It is noteworthy that the etiology and pathogenesis of primary open-angle glaucoma (POAG) have been not fully elucidated despite numerous studies. Currently, vascular endothelial dysfunction leading to optic disc ischemia and compression of optic nerve axons is a major current theory for the development of POAG [9-13]. Inadequate blood supply to the retina and optic nerve due to poor ocular circulation are important factors of the development and progression of POAG [14-16]. It has been demonstrated in recent years that a healthy endothelium regulates vascular tone capacity, modulates homeostasis, impacts vascular permeability, and regulates vessel growth [10, 17-20]. Damaged or excessively activated endothelial cells can also secrete vasoconstrictor factors, the best known of which is endothelin-1 (ET-1), as well as factors that affect the differentiation and growth of vascular smooth muscle cells. Endothelial cells also interact with cells in the bloodstream, ET-1 and other factors are released from endothelial cells into the bloodstream, where their chemotactic action can induce leucocytes and platelets to migrate to the endothelial wall [6, 21, 22]. Endothelial cells induce adhesion by expression of specific surface adhesion molecules (selectins, integrins and a supergene family of immunoglobulins) that can interact with ligands on the leucocytes and platelets. The disturbed permeability of the endothelial layer in patients with diabetes mellitus and/or hyperlipidaemia leads to an increased influx of substances from the circulation into the vessel wall. Therefore, endothelial dysfunction implies an imbalance between relaxation and contraction, between procoagulation and anticoagulation, and between proinflammatory and anti-inflammatory mediators, and may play a substantial role in the pathogenesis of POAG [23-25]. Because an increased production of superoxide anions in the vascular wall contributes to endothelial dysfunction in POAG, it is important to investigate the factors contributing to increased endothelial permeability in patients with POAG. The analysis of the literature on endothelial dysfunction demonstrated that the problem is still relevant and the mechanism of the development of endothelial dysfunction in POAG is still poorly understood [20, 26-29]. The purpose of this study was to investigate the value of the factors affecting vascular endothelium (oxidative stress, complement components and blood lipid profle) in patients with primary open-angle glaucoma. Material and Methods Ninety two patients (40 men and 22 women; age, 65 to 80 years) with POAG and 30 controls (somatically healthy individuals of a similar age and no known eye disease) were included in the study. Exclusion criteria were the presence of any other eye disease (excepting mild cataract), refractive error (myopia, hyperopia exceeding 6.0 D, and astigmatism exceeding 3.0 D), chronic autoimmune disease, diabetes mellitus, systemic disease, history of acute circulatory disorder, cancer or infection disease. Only patients on medication in the form of a single latanoprost dose and having no history of eye surgery were included in the analysis. All patients were seen regularly by a neurologist. Doppler evaluation of brachycephalic vessels was performed to exclude abnormality in these vessels. Patients had their systemic medications influencing platelet homeostasis and blood pressure discontinued by the time of the study. Patients with history of symptoms of primary or secondary vascular dysregulation (migraine, Raynaud syndrome, vasospasm, or neurocirculatory dystonia) were excluded from the controls. General clinical and special methods of examination were employed. Patients underwent an eye examination including visual acuity, Maklakoff tonometry, non-contact tonometry, biomicroscopy, gonioscopy, and pachymetry. A fasting venous blood sample was collected from them, from the median cubital vein, and plasma was separated by centrifugation at 2000 rev/min for 15 minutes. Lipid peroxidation characteristics, particularly, malondialdehyde (MDA) levels, were measured to assess oxidative stress. Plasma MDA levels were determined using thiobarbituric acid colorimetry with TBK-AGAT assays (Agat-Med, Russia). The activity of superoxide dismutase (SOD) was determined based on the inhibition of adrenaline autooxidation in alkaline medium by the enzyme due to the dismutation of superoxide anion radicals. Catalase activity was determined spectrophotometrically, based on the ability of hydrogen peroxide to form the stable colored complex with molybdenum salts. Standard biochemical techniques were used to determine complete blood count, clinical urinalysis, cholesterol, triglycerides, low-density lipoproteins (LDLs), and high-density lipoproteins (HDLs). Hypertriglyceridemia was defined as triglycerides ≥ 150 mg/dL (≥ 1.7 mmol/L). Serum C3 and C5 levels were determined using assays produced by Human company (Wiesbaden, Germany). Microsoft Excel 2010 and SPSS16.0 for Windows software and variation statistics were used for statistical analysis. Mean, standard error of mean (SEM), and standard deviation (SD) values were calculated. Student t test was employed to assess difference between the groups. The level of significance p < 0.05 was assumed. Results Table 1 shows plasma lipid peroxidation and antioxidant activity characteristics in patients with POAG and controls. Plasma MDA levels were 87% higher in patients than in controls (Table 1). Plasma SOD activity levels were 53.2% lower, and catalase activity levels, 240% higher in patients than in controls.
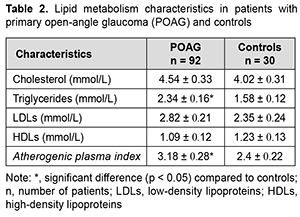
Table 2 shows plasma lipid metabolism levels in patients with POAG and controls. There was no significant difference (p>0.05) in plasma cholesterol or LDL levels between patients and controls. In addition, plasma triglycerides levels were 1.5 times higher, atherogenic plasma index, 1.3 times higher, and plasma HDL levels, 11.4% lower (p<0.05) in patients with POAG than in controls (Table 2).
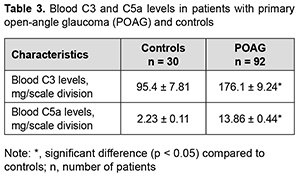
Table 3 shows blood C3 and C5a levels in patients with POAG and controls. Blood C3 levels were 1.9 times higher, and blood C5a levels, 6 times higher in patients with POAG than in controls (Table 3).
C5a function is enabled by the fixation of C3b. When C3b is initialized, the entire complement activation pathway progresses through any of the pathways, resulting in the formation of anaphylactic C5a and the terminal membrane-attacking complex C5B-9. Accordingly, high C5a levels found in the plasma of patients with POAG are a predisposing factor for the formation of autoaggression to the vascular endothelium. Consequently, an increase in the levels of this sub-component potentiates the damaging effect of membrane-attacking complexes, leading to vascular endothelial dysfunction. Discussion The results obtained indicate increased lipid peroxidation (namely, the presence of apparent oxidative stress in plasma) in patients with POAG. In our opinion, lipoproteins and nonesterified fatty acids are the major targets of lipid peroxidation in plasma. Our findings of increased lipid peroxidation in plasma of patients with POAG are in agreement with the reports of others that oxidative stress is an important factor of risk for endothelial dysfunction [14, 19, 30]. Lipid peroxidation is a physiological process continuously running in living cells. However, an excessive amount of active oxygen species triggers a cascade of reactions that results in destabilization and abnormal barrier functions in biological membranes. These biologically active species affect not only vascular tonicity, but also vascular structure and permeability. Oxidative stress as a mechanism of injury leading to degeneration of the subcellular components in retinal ganglion cells can act through signaling mechanisms, resulting in retinal cell death. In addition, MDA produced by lipid peroxidation can cause cross-linking and polymerization of membrane components. This can alter intrinsic membrane properties such as deformability, ion transport, enzyme and receptor activity, the aggregation state of cell surface determinants, etc. The function of numerous plasma proteins can be also affected by lipid peroxidation [12]. Of note that an increased plasma catalase activity is likely to be caused by catalase release from cells due to cell damage or destruction in the presence of oxidative stress. Consequently, while preserving its activity, catalase may in some way contribute to the compensation of oxidative stress in plasma. However, as we have reported previously, patients with POAG demonstrated intensive lipid peroxidation processes in spite of increased activity of catalase [27, 28]. Kovacevic and colleagues [2] concluded that higher values of total cholesterol, particularly atherogenic LDL fraction, may have certain influence in glaucoma. Serum lipid values were similar in both groups for triglycerides, HDL and LDL lipoproteins but cholesterol values were significantly higher in the POAG group. In a study by Egorov and colleagues [3], biochemical analysis of plasma lipids in patients with POAG showed atherogenic hyperlipidemia with decreased antioxidant activity. Davari and colleagues [12] found a positive association between POAG and dyslipidemia (OR=7.14 [95% CI: 2.3-22.2] for hypercholesterolemia and OR=16.9 [95% CI: 2.1-14.8] for hypertriglyceridemia. They concluded that hyperlipidemia can be a risk factor for POAG. In a similar study in 2009, by Pavljasevic and Asceric [31] in Bosnia and Herzegovina, the researchers tested 50 patients with open-angle glaucoma and 50 healthy individuals with respect to their serum lipids. The cholesterol mean value in the test group was 6.14 mol/dm whereas in the control group it was 5.96 mol/dm. The triglyceride mean value in the test group was 2.38 mol/dm and in the control group it was 2.04 mol/dm. High density cholesterol was average in the test group with 1.45 mol/dm and in the control group 1.40 mol/dm. Low density cholesterol in the test group was 3,98 mol/m and in the control group 4.08 mol/m. This means that blood cholesterol levels for patients in the test group were higher compared to those of the control group and could suggest that hypercholesterolemia could be one of predictable factor in POAG diagnosis [31]. In the Beijing eye study [25], about 3251 individuals (age > 45 years) had their complete ophthalmic examination. Blood serum lipids were also measured. The effect of dyslipidemia on the incidence of ophthalmic diseases was studied after adjustment for various factors (such as age, sex, residence, income level, body mass index, cigarette smoking, diastolic blood pressure, and blood sugar). Results showed that IOP was significantly increased in dyslipidemic patients. Recent studies have noted that high-density lipoproteins (a major anti- atherogenic lipoprotein fraction) protect endothelial cells from apoptosis. HDLs also block the mitochondrial pathway of apoptosis by inhibiting dissipation of mitochondrial potential, generation of reactive oxygen species, and release of cytochrome c into the cytoplasm. As a consequence, HDLs prevent activation of caspases 9 and 3 and apoptotic alterations of the plasma membrane such as increase of permeability and translocation of phosphatidylserine. Studies identified HDL as a carrier of endogenous endothelial survival factors, thus confirming previous findings [15, 20, 32]. Three major components are recognized as the major contributing factors in endothelial reperfusion injury. These are: (1) molecular oxygen; (2) cellular blood elements (especially the neutrophils); and (3) components of the activated complement system, notably C5a, which are potent stimulators of neutrophil superoxide production [11]. Complement fragments such as the anaphylatoxins C3a and C5a, are produced both locally and systematically, and the membrane attack complex is deposited on cell membranes and subsequent release of mediators such as histamine, thereby causing an increase in vascular permeability with concomitant manifestation of cellular edema. Proteolytic complement fragments (C3a, C4a, and C5a) induce acute inflammation by activating mast cells, neutrophils and endothelial cells, and are a cause of endothelial dysfunction. All three peptides bind to mast cells and induce degranulation, with the release of vasoactive mediators such as histamine. In neutrophils, C5a stimulates motility, firm adhesion to endothelial cells, and, at high doses, stimulation of the respiratory burst and production of reactive oxygen species. In addition, C5a may act directly on vascular endothelial cells and induce increased vascular permeability and the expression of P-selection, which promotes neutrophil binding. This combination of C5a actions on mast cells, neutrophils, and endothelial cells contributes to inflammation at sites of complement activation. C5a is the most potent anaphylatoxin. In addition, it is a chemotactic factor for neutrophils and stimulates the respiratory burst in them and it stimulates inflammatory cytokine production by macrophages. It causes induction of adhesion molecules on endothelial cells, resulting in increased vascular permeability and contraction of smooth muscle cells [10]. C3a and C5a bind to mast cells and cause degranulation, with the release of vasoactive mediators such as histamine, thereby causing an increase in vascular permeability with concomitant manifestation of cellular edema. In our opinion, the elevated C5a levels we found in the plasma of patients with POAG are a predisposing factor to the formation of autoaggression against the vascular endothelium, increased vascular endothelial permeability and eventual death of retinal cells. Conclusion First, blood MDA levels in patients with POAG were 87% higher compared to controls. In addition, plasma SOD activity levels were 53.2% lower, and catalase activity levels, 240% higher in patients compared to controls. Moreover, C3 and C5 levels in the blood were 1.9 times and 6 times, respectively, higher in patients with POAG than in controls. Second, we found an imbalance between the prooxidant and antioxidant systems, abnormal composition of blood lipids (e.g., hypercholesterolemia and hypertriglyceridemia) and elevated C5a levels in patients with POAG.
References 1.Jiang F, Guo Y, Salvemini D, Dusting GJ. Superoxide dismutase mimetic M40403 improves endothelial function in apolipoprotein(E)-deficient mice. Br J Pharmacol. 2003;139(6):1127-34. 2.Kovacevic S, Jurin A, Didovic-Torbarina A. Dislipidmija u bolesnikas a Primarnim glaukomomotvorenogugla. Abstracts of the 7th Congress of the Croatian Ophthalmol. Society with International Participation. Ophthalmol Croatica. 2007; 16:51. 3.McGwin G Jr, McNeal S, Owsley C, Girkin C, Epstein D, Lee PP. Statins and others cholesterol-lowering medications and the presence of glaucoma. Arch Ophthalmol. 2004;122:822-6. 4.Tuychibaeva D, Rizaev J, Malinouskaya I. [Dynamics of primary and general incidence due to glaucoma among the adult population of Uzbekistan]. Oftalmologiia. Vostochnaia Ievropa. 2021;11.1:27–38. Russian. 5.Tuychibaeva DM. [Main Characteristics of the Dynamics of Disability Due to Glaucoma in Uzbekistan]. Oftalmologiia. Vostochnaia Ievropa. 2022;12.2:195-204. Russian. 6.Rizaev J, Tuychibaeva D. Study of the general state and dynamics of primary and general disability due to glaucoma of the adults in the republic of Uzbekistan and the city of Tashkent. Journal of Dentistry and Craniofacial Research. 2022;1(2):75–7. 7.Тuychibaeva DM. Longitudinal changes in the disability due to glaucoma in Uzbekistan. J Ophthalmol (Ukraine). 2022;507.4:12-17. 8.Tuychibaeva DM, Rizayev JA., Stozharova NK. Longitudinal changes in the incidence of glaucoma in Uzbekistan. J Ophthalmol (Ukraine). 2021;4:43-7. 9.Vlasova SP, Ilchenko MIu, Kazakova EB, et al. [Endothelial dysfunction and arterial hypertension]. Samara: Ofort; 2010. Russian. 10.Egorov EA, Bachaldin IL, Sorokin EL. [Characteristics of morphological and functional state of erythrocytes in patients with primary open-angle glaucoma with normalized intraocular pressure]. Vestn Oftalmol. 2001 Mar-Apr;117(2):5-8. 11.Irtegova EIu. [Role of vascular endothelial dysfunction and ocular blood flow in the development of glaucomatous optic neuropathy]. [Thesis for the degree of Cand Sc (Med)]. Moscow, 2015. Russian. 12.Félétou M, Vanhoutte PM. Endothelial dysfunction: a multifaceted disorder (The Wiggers Award Lecture). Am J Physiol Heart Circ Physiol. 2006 Sep;291(3):H985-1002. 13.Flammer J, Konieczka K. Retinal venous pressure: the role of endothelin. EPMA J. 2015 Oct 26; 6:21. 14.Malishevskaia TN, Kiseleva TN, Filippova IuE, et al. [Antioxidant status and blood lipid profile in patients with various courses of primary open-angle glaucoma]. Oftalmologiia. 2020;17(4):761-70. Russian. 15.Stangeby DK, Ethier CR. (September 17, 2001). Computational Analysis of Coupled Blood-Wall Arterial LDL Transport. J Biomech Eng. 2002 Feb;124(1):1–8. 16.Tezel G. Oxidative stress in glaucomatous neurodegeneration: mechanisms and consequences. Prog Retin Eye Res.2006;25:490-513 17.Bachaldin IL. [Role of blood rheology abnormalities in the progression of open-angle glaucoma with normalized intraocular pressure and developing principles for treatment of the disease]. [Thesis for the degree of Cand Sc (Med)]. Khabarovsk, 2004. Russian. 18.Lyndina ML, Shishkin AN. [Clinical features of endothelial dysfunction in obesity and the role of smoking factor]. Regionarnoie krovoobrashcheniie i mikrotsirkuliatsiia. 2018;17(2):20—7. Russian. 19.Gubin DG, Malishevskaya TN, Astakhov YS, Astakhov SY, Kuznetsov VA, Cornelissen G, Weinert D. Progressive retinal cell loss in primary open-angle glaucoma is associated with temperature circadian rhythm phase delay and compromised sleep. Chronobiology International. 2019;36(4):564-77. 20.Nofer JR, Levkau B, Wolinska I, Junker R, Fobker M, von Eckardstein A, et al. Suppression of endothelial cell apoptosis by high density lipoproteins (HDL) and HDL-associated lysosphingolipids. J Biol Chem. 2001 Sep 14;276(37):34480-5. 21.Mikheytseva IN. [Pathogenetic importance of endothelial dysfunction in primary glaucoma]. Dosiagnennia biologii ta medytsyny. 2009; 14(2):17-20. Russian. 22.McMonnies C. Reactive oxygen species, oxidative stress, glaucoma and hyperbaric oxygen therapy. J Optom. 2018 Jan-Mar;11(1):3-9. 23.Fujino Y, Asaoka R, Murata H. Evaluation of Glaucoma Progression in Large-Scale Clinical Data: The Japanese Archive of Multicentral Databases in Glaucoma (JAM-DIG). Invest Ophthalmol Vis Sci. 2016 Apr1;57(4):2012-20.Crossref PubMed 24.Haefliger IO, Flammer J, Beny JL, Luscher TF. Endothelium-dependent vasoactive modulation in the ophthalmic circulation. Prog Retin Eye Res. 2001;20(2):209-25. 25.Wang S, Xu L, Jonas JB, You QS, Wang YX, Yang H. Dyslipidemia and eye diseases in the adult Chinese population: The Beijing eye study. PLoS One. 2011; 6: e26871. 26.Cellini M, Strobbe E, Gizzi C, Balducci N, Toschi PG, Campos EC. Endothelin-1 plasma levels and vascular endothelial dysfunction in primary open angle glaucoma. Life Sci. 2012 Oct 15;91(13-14):699-702. 27.Davari MH, Kazemi T, Rezai A. A survey of the relationship between serum cholesterol and triglyceride to glaucoma: A case control study. J Basic Appl Sci. 2014;10:39–43. 28.Emre M, Orgul S, Haufschild T, Shaw SG, Flammer J. Increased plasma endothelin-1 levels in patients with progressive open angle glaucoma. Br J Ophthalmol. 2005 Jan;89(1):60-3. 29.Fadini GP, Pagano C, Baesso I, Kotsafti O, Doro D, de Kreutzenberg S. V, et al. Reduced endothelial progenitor cells and brachial artery flow-mediated dilation as evidence of endothelial dysfunction in ocular hypertension and primary open-angle glaucoma. Acta Ophthalmol. 2010 Feb. 88(1):135-41. 30.Kurysheva NI, Irtegova EIu, Iasamanova AN, Kiseleva TN. [Endothelial dysfunction and platelet hemostasis in primary open-angle glaucoma]. Natsional'nyi zhurnal Glaucoma. 2015;14(1):27-36. Russian. 31.Pavljasevic S, Asceric M. Primary open-angle glaucoma and serum lipids. Bosn J Basic Med Sci. 2009 Feb; 9(1):85-8. 32.Jensen JS, Feldt-Rasmussen B, Jensen KS, Clausen P, Henrik Scharling H, Nordestgaard BG. Transendothelial lipoprotein exchange and microalbuminuria. Cardiovasc Res. 2004 Jul 1;63(1):149-54.
Information about authors and disclosure of information Corresponding author: Tuychibaeva D.M. – dyly@mail.ru. Conflict of interest: The authors have declared that no competing interests exist. Ethical approval: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Authors fulfilled the ICMJE authorship criteria. Funding information: This research does not received specific grant from any funding agency in the public, commercial or not-for -profit sector.
|