J.ophthalmol.(Ukraine).2022;4:33-39.
|
http://doi.org/10.31288/oftalmolzh202243339 Received: 12.06.2022; Accepted: 05.08.2022; Published on-line: 24.08.2022 Effects of carnosine on the pro-antioxidative status of the anterior segment of the eye in experimental uveitis plus ocular hypertension I. M. Mikheytseva, N. V. Bondarenko, S. G. Kolomiichuk SI "The Filatov Institute of Eye Deseases and Tissue Therapy of the NAMS of Ukraine"; Odesa (Ukraine) TO CITE THIS ARTICLE: Mikheytseva IM, Bondarenko NV, Kolomiichuk SG. Effects of carnosine on the pro-antioxidative status of the anterior segment in experimental uveitis plus ocular hypertension. J.ophthalmol.(Ukraine).2022;4:33-39. http://doi.org/10.31288/oftalmolzh202243339
Background: Finding means for improving the effectiveness of drug therapy of ocular inflammatory disorders, especially those with comorbidities, is a challenge to ophthalmology. Purpose: To assess the efficacy of carnosine for improving the pro/antioxidative imbalance in the anterior segment of rabbit eyes with experimental anterior uveitis in the presence of ocular hypertension (OHT). Material and Methods: Thirty-three rabbits were used in the study. Group 1 (the control group) comprised 9 intact rabbits and group 2, 12 animals with OHT induced prior to experimental non-infectious uveitis. Group 3 comprised 12 animals in which carnosine 5% was administered bilaterally into the conjunctival sac twice daily for four weeks for OHT induced prior to experimental non-infectious uveitis. Non-infectious uveitis was induced by injecting albumin into the anterior chamber, and OHT was induced by a single 0.1-mL injection of carbomer 0.3% into the anterior chamber. Activities of superoxide dismutase, catalase, glutathione peroxidase, NADH-oxidase and xanthine oxidase, and levels of total proteins and malondialdehyde (MDA) and diene conjugate (DC) in uveal tissue (iris and ciliary body) and aqueous humor specimens were determined. Results: Uveal activities of NADH-oxidase and xanthine oxidase were 19.4% (р < 0.05) and 27.2% (р < 0.01), respectively, lower in eyes of rabbits treated with carnosine than in eyes of animals not treated for anterior uveitis plus OHT. Uveal activities of glutathione peroxidase, superoxide dismutase and catalase were 34.7%, 39.6% and 28.4%, respectively, higher, and uveal MDA and DC levels were 39.3% and 33.3%, respectively, lower, in eyes of the former rabbits than of the latter rabbits. Similar changes were found in aqueous humor specimens of the experimental rabbits. Conclusion: Carnosine, when instilled locally, contributed to improvement in the pro/antioxidative imbalance in the anterior segment of rabbit eyes with uveitis plus OHT. It is reasonable to include carnosine-based eye drops into multiagent therapy of uveitis complicated by OHT due to their antioxidative, anti-inflammatory and membrane-stabilizing effects. Keywords: non-infectious uveitis, ocular hypertension, antioxidative enzymes, peroxidation, carnosine
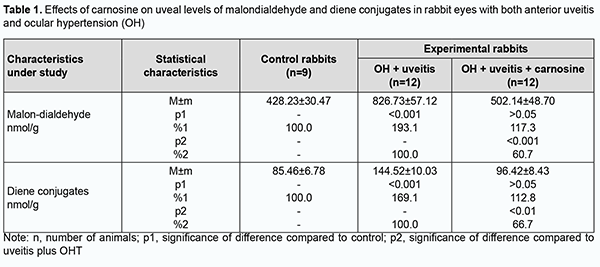
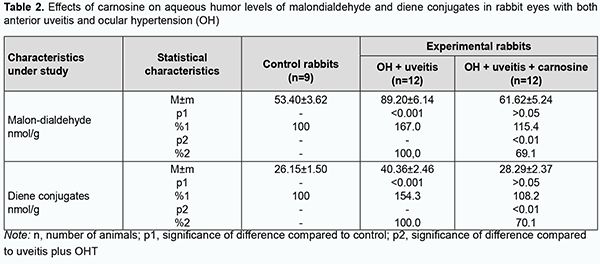
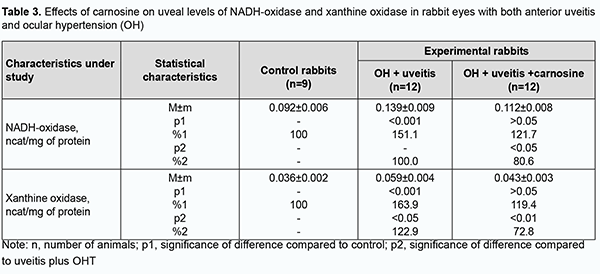
Introduction Ocular inflammation can result in reduced visual acuity and even blindness, and affects functional work capacity and quality of life, posing a social and economic challenge for the society. Therefore, finding means for improving the effectiveness of drug therapy of ocular inflammatory disorders, especially those with comorbidities, is a challenge for clinical ophthalmologists [1,2]. Etiotropic and pathogenetic therapy for uveitic inflammatory processes involves immunosuppressive, steroidal and non-steroidal anti-inflammatory agents [2-4]. Corticosteroids and non-steroidal anti-inflammatory agents, although adequately effective, can result in metabolic abnormalities or even glaucoma and cataract, when applied for a long period [5]. Therefore, it is reasonable to study the efficacy of agents with other mechanisms of action to reduce the incidence and severity of systemic complications and local ocular complications. The use of biologic agents like tumor necrosis factor alpha inhibitors (anti-TNFα) in the treatment of uveitis has been studied [2, 6-8]. Of note that increased oxidative and peroxidative processes result in oxidative balance disturbance and are a pathogenetic factor in ocular inflammatory and degenerative processes, especially in the presence of raised intraocular pressure (IOP) [9-16]. Therefore, it is reasonable to include antioxidative agents (lycopene [17], N-acetylcysteine [15], betaine [18], and others [9, 16, 19]) normalizing ocular metabolic abnormalities in the list of pathogenetic treatment for the above disorders. We have previously found that oxidative stress with a suppressed antioxidative status in the eye is a pathogenetic factor which in the presence of raised IOP contributes to increased severity of experimental uveitis [13, 14, 20]. Therefore, we decided to explore the potential of dipeptide carnosine (beta-alanyl-L-histidine), an antioxidative agent, in the treatment of experimental non-infectious uveitis in the presence of raised IOP. This biologically active substance has been proposed for the prevention of chronic disorders. The naturally occurring dipeptide carnosine is a highly bioavailable low-molecular hydrophilic antioxidant that is capable of crossing the blood-brain barrier and contributing to membrane stabilization [21]. It is believed that carnosine, a dipeptide with anti-inflammatory, antioxidative and antiglycating properties may have a role in the prevention of chronic diseases [22]. The purpose of the study was to assess the efficacy of carnosine for improving the pro/antioxidative imbalance in the anterior segment of rabbit eyes with experimental anterior uveitis in the presence of ocular hypertension (OHT). Material and Methods All animal experiments were performed in compliance with the General Ethical Principles of Animal Experiments (approved by Third National Congress on Bioethics, Ukraine, Kyiv, 2007) and European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes from the European Treaty Series (Strasbourg, 1986). Thirty-three Chinchilla rabbits maintained under normal vivarium conditions and fed and watered ad libitum, were used for all experiments. Group 1 (the control group) comprised 9 intact rabbits and group 2, 12 animals with OHT induced prior to experimental non-infectious uveitis. Group 3 comprised 12 animals in which beta-alanyl-L-histidine (carnosine) 5% was administered bilaterally into the conjunctival sac twice daily for four weeks for OHT induced prior to experimental non-infectious uveitis. Non-infectious uveitis was induced by introducing bovine serum albumin into the anterior chamber [23], and OHT was induced by a single 0.1-mL injection of carbomer 0.3% into the anterior chamber [24]. We used a Maklakoff tonometer with a 7.5-g plunger load to perform IOP measurements after topical anesthesia was performed by instillation of proparacaine hydrochloride 0.5% into the conjunctival sac. Biomicroscopy was used to ascertain the state of the anterior segment. Four weeks after completing OHT plus uveitis modeling, animals were deeply anesthetized with thiopental sodium 10% (1.0 mL/kg, intramuscularly) and euthanized by air embolism. Eyes were enucleated and immediately placed on ice at 0 to 5 ºC. Uveal tissues (specifically, the iris and ciliary body) were used to prepare the homogenate with NaCl 0.9% at a 1:9 mass to volume ratio. Samples underwent centrifugation at 10,000 revolution per minute for 10 minutes at 5 ºС and the supernatant was used for biochemical analysis. The activities of antioxidant enzymes, superoxide dismutase [25], catalase [26] and glutathione peroxidase [27], xanthine oxidase [28], and the levels of total proteins [29] and lipid peroxidation products, malondialdehyde and diene conjugate [30] in the supernatant were determined. The mitochondrial precipitate of differential centrifugation of tissue homogenate was resuspended to determine the activity of NADH-oxidase [31] in insulated mitochondria of the uveal tissue. Statistica 5.5 (StatSoft, Tulsa, OK, USA) software and parametric statistical tests were used for statistical analysis. Results Increased lipid peroxidation (Tables 1, 2) and increased activities of NADH-oxidase and xanthine oxidase (the enzymes producing active oxygen species) (Table 3) were observed in the uveal tissue and aqueous specimens of rabbits with experimental non-infectious uveitis in the presence of OHT.
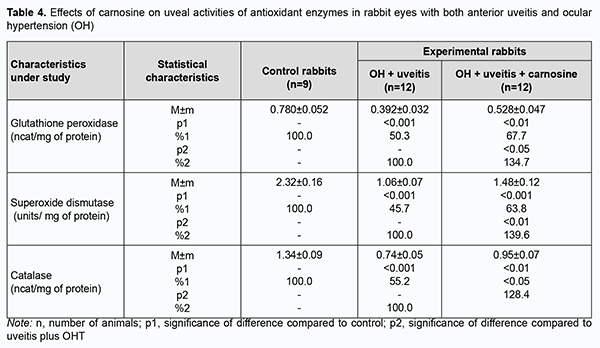
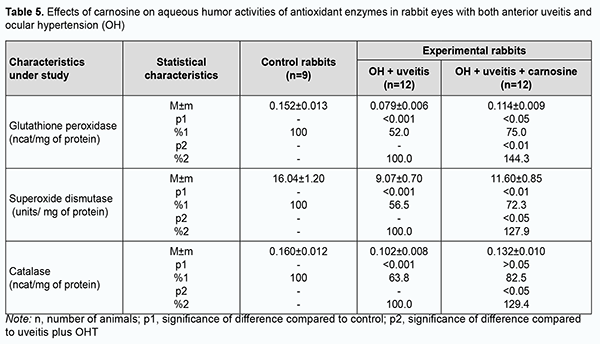
Aside from accumulation of lipid peroxidation products and free radical species in the eye, the above processes resulted in exhaustion of the antioxidative enzyme system. The activities of antioxidant enzymes, superoxide dismutase, catalase and glutathione peroxidase (Tables 4, 5) in these animals were reduced compared to controls.
We have previously reported that binocular instillation of carnosine solution in the conjunctival sac for 4 weeks resulted in a significant decrease in the severity of inflammation in the anterior and posterior segments, leading to an improved clinical picture with an improvement in uveal metabolism, in eyes of the animals treated for experimental allergic uveitis in the presence of OHT [20]. In the current study, uveal malondialdehyde and diene conjugate levels were 39.3% and 33.3%, respectively, lower in eyes of rabbits treated with carnosine than in eyes of animals not treated for anterior uveitis plus OHT, and these differences were statistically significant. In addition, malondialdehyde and diene conjugate levels in aqueous humor specimens were 30.9% and 29.9%, respectively, lower for the former rabbits than for the latter rabbits (Tables 1, 2). The differences compared with controls were, however, not statistically significant. Table 3 presents the data on the effects of carnosine on uveal activities of NADH-oxidase and xanthine oxidase in rabbit eyes with anterior uveitis plus OHT. Uveal activities of NADH-oxidase and xanthine oxidase were 21.9% and 19.2%, respectively, higher in eyes of rabbits treated with carnosine for anterior uveitis plus OHT than in eyes of the controls, but these differences were not statistically significant. Moreover, uveal activities of NADH-oxidase and xanthine oxidase were 19.4% (р < 0.05) and 27.2% (р < 0.01), respectively, lower in eyes of rabbits treated with carnosine than in eyes of animals not treated for anterior uveitis plus OHT. Therefore, carnosine, when applied locally for experimental non-infectious uveitis plus OHT for a long time, contributed to a reduction in (a) the accumulation of lipid peroxidation products and (b) the opportunity for free-radical production in the presence of increased activity of pro-oxidative enzymes, in tissues of the anterior segment of the eye. Of note is an apparent activating effect of carnosine on anti-oxidative enzymes in the uveal tissue of animals with uveitis plus OHT (Table 4). Uveal activities of glutathione peroxidase, superoxide dismutase and catalase were 34.7% (р < 0.05), 39.6% (р < 0.01) and 28.4% (р < 0.05) respectively, higher in eyes of rabbits treated with carnosine than in eyes of animals not treated for anterior uveitis plus OHT (Table 4). However, uveal activities of glutathione peroxidase, superoxide dismutase and catalase were 32.3% (р < 0.01), 36.2% (р < 0.01) and 29.1% (р < 0.01) respectively, lower in eyes of rabbits treated with carnosine for anterior uveitis plus OHT than in eyes of the controls. Aqueous humor activities of glutathione peroxidase, superoxide dismutase and catalase were 44.3% (р < 0.01), 27.9% (р < 0.05) and 29.4% (р < 0.05), respectively, higher in eyes of rabbits treated with carnosine than in eyes of animals not treated for anterior uveitis plus OHT (Table 5), and 25.0% (р < 0.05), 27.7% (р < 0.01), and 17.5% (р > 0.05), respectively, lower in eyes of the former animals than in eyes of the controls. In summary, although activities of antioxidative enzymes in the anterior segment were somewhat lower for rabbits treated with carnosine for anterior uveitis plus OHT than for the controls, our findings demonstrated apparent beneficial effects of carnosine eye drops on the improvements in (a) the activities of these enzymes and (b) the pro/antioxidative imbalance in the ocular tissues of the former animals. Discussion Pathogenetic features of ocular inflammatory and degenerative disorders (e.g., non-infectious uveitis complicated by ocular hypertension) indicate that free-radical process activation in the presence of exhaustion of the antioxidative system in uveal tissues is an important trigger of metabolic abnormalities and functional and structural alterations in the eye [11, 12, 15, 20, 32]. That is why we aimed to investigate the effects of dipeptide carnosine, an antioxidative agent [33-36], on the enzymatic antioxidative system, activities of the enzymes producing active oxygen species, and levels of peroxidation products in the anterior segment of the rabbit eye in experimental uveitis plus OHT. We demonstrated the capacity of carnosine to reduce the uveal oxidative stress induced by ocular inflammation and complicated by raised IOP. The carnosine molecule, the main component responsible for its physiological and biochemical properties, is the imidazole part of l-Histidine[34]. The biochemical properties of carnosine [34] include metal-ion chelation [37] and the capacity of reacting with carbonyls [38], which is important under carbonyl stress conditions in various disorders. Thus, carnosine can (a) react with various aldehydes (like methylglyoxal) with a reduction in glycation damage and (b) suppress the deleterious effects of protein carbonyls by reacting with them to form protein-carbonyl-carnosine adducts [35, 39-41]. In an in vivo study by Aydin and colleagues [42], administration of carnosine for 1 month to aged rats resulted in decreased levels of lipid peroxidation products in serum and increased levels of reduced glutathione in erythrocytes. Therefore, carnosine and its derivatives are believed to be promising for the prevention of complications in various pathological conditions caused by oxidative stress [32, 33, 35, 36]. In a study by Tsai and colleagues [43], mice treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) were used to examine the neuroprotective effects of carnosine. The preintake of carnosine significantly attenuated MPTP-induced glutathione loss, retained the activity of antioxidative enzymes, diminished oxidative stress, and lowered inflammatory cytokines. In the current study, carnosine caused a reduction in malondialdehyde and diene conjugate levels and activities of NADH-oxidase and xanthine oxidase in the anterior segment of the rabbit eye in experimental non-infectious uveitis plus OHT. In addition, carnosine contributed to a substantial increase in uveal and aqueous humor activities of glutathione peroxidase, superoxide dismutase and catalase in experimental anterior uveitis plus OHT in rabbits. Moreover, we have previously reported that carnosine instillation in the conjunctival sac signiticantly reduced neopterin levels in uveal tract tissue, aqueous humor and tear samples from the rabbit eye with experimental anterior uveitis plus OHT, indicating a reduction in inflammation in the anterior segment of the eye [20, 44]. Therefore, we demonstrated that carnosine, when instilled in the eye for a long time, contributed to improvement in the pro/antioxidative imbalance in the anterior segment of the rabbit eye with experimental non-infectious uveitis plus OHT. Consequently, we believe it is reasonable to include antioxidative agents into multiagent therapy of non-infectious uveitis, especially that complicated by OHT, for preventing metabolic abnormalities in the anterior segment of the eye. Carnosine-based eye drops would be the first-line therapy in this situation due to their antioxidative, anti-inflammatory and membrane-stabilizing effects.
References 1.Zborovskaya AV, Dorokhova A.V. [Use of biological therapy in the treatment of uveitides: Current trends]. Oftalmol Zh. 2017;5:60-5. Russian. 2.Krakhmaleva DA, Pivin EA, Trufanov SV, Malozhen SA. [Modern opportunities in uveitis treatment]. Ophthalmology in Russia. 2017;14(2):113-119. Russian. 3.Relhan N, Yeh S, Albini TA. Intraocular sustained-release steroids for uveitis. Int Ophthalmol Clin. 2015; 55(3): 25–38. 4.Jabs DA. Immunosuppression for the Uveitides. Ophthalmology. 2018;125:193–202. 5.Friedman DS, Holbrook JT, Ansari H, et al. Risk of elevated intraocular pressure and glau coma in patients with uveitis: results of the multicenter uveitis steroid treatment trial. Ophthalmology. 2013 Aug;120(8):1571-9. 6.Gallego-Pinazo R, Dolz-Marco R, Martínez-Castillo S, et al. Update on the principles and novel local and systemic therapies for the treatment of non-infectious uveitis. Inflamm Allergy Drug Targets. 2013 Feb;12(1):38-45. 7.Panchenko NV, Khramova TA, Litvishchenko AI, et al. [Changes in the basal vitreous in uveitis treated by adalimumab]. Oftalmol Zh. 2015;6:64-7. Russian. 8.Lerman MA, Rabinovich CE. The Future Is Now: Biologics for Non-Infectious Pediatric Anterior Uveitis. Paediatr Drugs. 2015 Aug;17(4):283-301. 9.Yadav UC, Kalariya NM, Ramana KV. Emerging role of antioxidants in the protection of uveitis complications. Curr Med Chem. 2011;18(6):931-42. 10.Njie-Mbye YF, Kulkarni-Chitnis M, Opere CA, et al. Lipid peroxidation: pathophysiological and pharmacological implications in the eye. Front Physiol. 2013 Dec 16;4:366. 11.Yadav UCS. Oxidative Stress-Induced Lipid Peroxidation: Role in Inflammation. In: Rani V, Yadav UCS, eds. Free Radicals in Human Health and Disease. New Delhi: Springer India; 2015. pp.119-29. 12.Nita M, Grzybowski A.The Role of the Reactive Oxygen Species and Oxidative Stress in the Pathomechanism of the Age-Related Ocular Diseases and Other Pathologies of the Anterior and Posterior Eye Segments in Adults. Oxid Medicine Cell Longev. 2016;2016:3164734. 13.Mikheytseva IM, Bondarenko NV, Kolomiichuk SG, Siroshtanenko TI. Oxidation and peroxidation in the uvea of the rabbit eyes with experimental uveitis and ocular hypertension. J Ophthalmol (Ukraine). 2019;2:55-60. 14.Mikheytseva IM, Bondarenko NV, Kolomiichuk SG, et al. The effect of high intraocular pressure on enzymatic antioxidant system of uveal tissues in rabbits with experimental allergic uveitis. J Ophthalmol (Ukraine). 2019;4:57-63. 15.Hsu S-M, Yang C-H, Teng Y-T, et al. Suppression of the Reactive Oxygen Response Alleviates Experimental Autoimmune Uveitis in Mice. Int J Mol Sci. 2020 May 5;21(9):3261. 16.Vitovska OP, Pichkur LD. Prospects of up-to-date antioxidants in the treatment of chronic eye diseases. J Ophthalmol (Ukraine). 2020;3:42-6. 17.Göncü T, Oğuz E, Sezen H, et al. Anti-inflammatory effect of lycopene on endotoxin-induced uveitis in rats. Arq Bras Oftalmol. Nov-Dec 2016;79(6):357-62. 18.Choi Y, Jung K, Kim HJ, et al. Attenuation of Experimental Autoimmune Uveitis in Lewis Rats by Betaine. Exp Neurobiol. 2021 Aug 31;30(4):308-317. 19.Chen S-J, Lin T-B, Peng H-Y, et al. Protective Effects of Fucoxanthin Dampen Pathogen-Associated Molecular Pattern (PAMP) Lipopolysaccharide-Induced Inflammatory Action and Elevated Intraocular Pressure by Activating Nrf2 Signaling and Generating Reactive Oxygen Species. Antioxidants (Basel). 2021 Jul 7;10(7):1092. 20.Mikheitseva IM, Bondarenko NV, Kolomiichuk SG, Siroshtanenko TI. Clinical picture of uveitis in the presence of ocular hypertension and improvement in disease course with dipeptide carnosine. J Ophthalmol (Ukraine). 2021;1:55-61. 21.Prokopieva VD, Yarygina EG, Bokhan NA, Ivanova SA. Use of Carnosine for Oxidative Stress Reduction in Different Pathologies. Oxid Med Cell Longev. 2016;2016:2939087. 22.Menon K, Mousa А, de Courten В. Effects of supplementation with carnosine and other histidine-containing dipeptides on chronic disease risk factors and outcomes: protocol for a systematic review of randomised controlled trials. BMJ Open. 2018 Mar 22;8(3):e020623. 23.Information Bulletin No. 19 issued 10.10.2019, based on Pat. of Ukraine №137,107; MPK (2019.01) А61К 9/00. [Method for inducing non-infectious uveitis in the presence of ocular hypertension]. Authors: Mikheytseva IM, Kolomiichuk SG, Bondarenko NV, Siroshtanenko TI. Owner: State Institution Filatov Institute of Eye Diseases and Tissue Therapy, NAMS of Ukraine. Ukrainian. 24.Xu Y, Chen Z, Song J. A study of experimental carbomer glaucoma and other experimental glaucoma in rabbits. Zhonghua Yan Ke Za Zhi. 2002 Mar;38(3):172-5. 25.Makarenko EV. [Complex determination of the activity of superoxide dismutase and glutathione reductase in erythrocytes of patients with chronic liver disease]. Lab Delo. 1988;11:48-50. Russian. 26.Koroliuk MA, Ivanova LI, Maĭorova IG, Tokarev VE. [A method of determining catalase activity]. Lab Delo. 1988;(1):16-8. Russian. 27.Model MA. [On the determination of the activity of glutathione peroxidase]. Voporosy meditsinskoi khimii. 1989;4:132-3. 28.Fried R, Fried L. Xanthin-Oxydase (Xanthin-Dehydrogenase). In: HU Bergmeyer. [Methods of enzymatic analysis]. Berlin: Аcademie Verlag, 1984. p. 625-9. German. 29.Larson E, Howlet В, Jagendorf А. Artificial reductant enhancement of the Lowry method for protein determination. Anal Biochem. 1986 Jun;155(2):243-8.Crossref 30.Orekhovich VN. [Current methods in biochemistry]. Moscow: Meditsina; 1977. Russian. 31.Rybalchenko VK. [Membrane structure and function]. Kyiv: Vyshcha shkola; 1988. Russian. 32.Babizhayev MA. Generation of reactive oxygen species in the anterior eye segment. synergistic codrugs of N-acetylcarnosine lubricant eye drops and mitochondria-targeted antioxidant act as a powerful therapeutic platform for the treatment of cataracts and primary open-angle glaucoma. BBA Clin. 2016 Apr 19;6:49-68. 33.Boldyrev AA, Stvolinsky SL, Fedorova TN, Suslina ZA. Carnosine as a natural antioxidant and geroprotector: from molecular mechanisms to clinical trials. Rejuvenation Res. 2010 Apr-Jun;13(2-3):156-8. 34.Boldyrev AA, Aldini G, Derave W. Physiology and Pathophysiology of Carnosine. Physiol Rev. 2013 Oct;93(4):1803-45. 35.Cararo JH, Streck EL, Schuck PF, Ferreira G da C. Carnosine and Related Peptides: Therapeutic Potential in Age-Related Disorders. Aging Dis. 2015 Oct 1;6(5):369-79. 36.Babizhayev MA. Biological activities of the natural imidazole-containing peptidomimetics N-acetylcarnosine, carcinine and L-carnosine in ophthalmic and skin care products. Life Sci. 2006 Apr 11;78(20):2343-57. 37.Baran EJ. Metal complexes of carnosine. Biochemistry (Mosc). 2000 Jul;65(7):789-97. 38.Vistoli G, de Maddis D, Straniero V, et al. Exploring the space of histidine containing dipeptides in search of novel efficient RCS sequestering agents. Eur J Med Chem. 2013 Aug;66:153-60. 39.Hipkiss AR. Aging, Proteotoxicity, Mitochondria, Glycation, NAD and Carnosine: Possible Inter-Relationships and Resolution of the Oxygen Paradox. Front Aging Neurosci. 2010 Mar 18;2:10. 40.Hipkiss AR. Carnosine and protein carbonyl groups: a possible relationship. Biochemistry (Mosc). 2000 Jul;65(7):771-8. 41.Pepper ED, Farrell MJ, Nord G, Finkel SE. Antiglycation effects of carnosine and other compounds on the long-term survival of Escherichia coli. Appl Environ Microbiol. 2010 Dec;76(24):7925-30. 42.Aydín AF, Küskü-Kiraz Z, Doğru-Abbasoğlu S, Uysal M. Effect of carnosine treatment on oxidative stress in serum, apoB-containing lipoproteins fraction and erythrocytes of aged rats. Pharmacol Rep. 2010 Jul-Aug;62(4):733-9. 43.Tsai SJ, Kuo WW, Liu WH, Yin MC. Antioxidative and anti-inflammatory protection from carnosine in the striatum of MPTP-treated mice. J Agric Food Chem. 2010 Nov 10;58(21):11510-6. 44.Mikheytseva IM, Bondarenko NV, Kolomiichuk SG, Kuryltsiv NB. Neopterin level in the anterior segment of the eye in induced uveitis with ocular hypertension treated by dipeptide carnosine. J Ophthalmol (Ukraine). 2021;2:64-70.
Disclosures Corresponding Author: Kolomiichuk Serhii, fiatovbiochem@ukr.net, Author Contribution: Miheytseva I.M. – concept; writing design, review and editing; Kolomiychuk S.H. – modeling of type II diabetes, writing. All authors analyzed the results and agreed on the final version of the manuscript. Conflict of Interests: The authors declare no conflict of interest that could influence their opinion regarding the subject matter or materials described and discussed in this manuscript. Sources of Support: None Disclaimer: the considerations presented in the article are exclusively the author's and do not represent the official position of the institution. Abbreviations: OHT – ocular hypertension; IOP –intraocular pressure; MDA – malondialdehyde; DC – diene conjugate
|