J.ophthalmol.(Ukraine).2020;6:30-37.
|
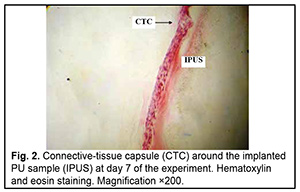
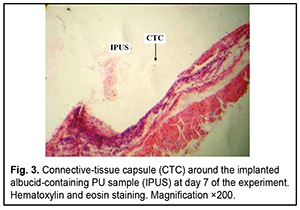
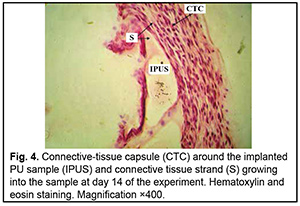
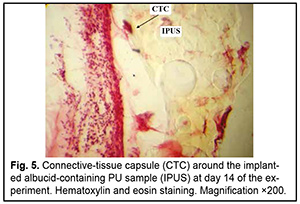
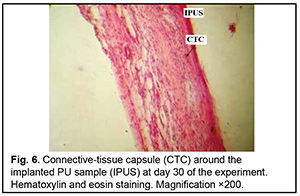
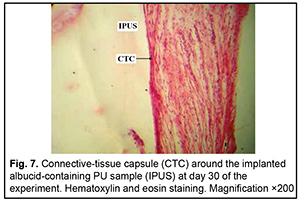
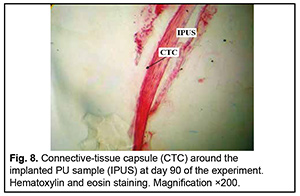
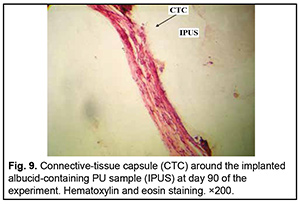
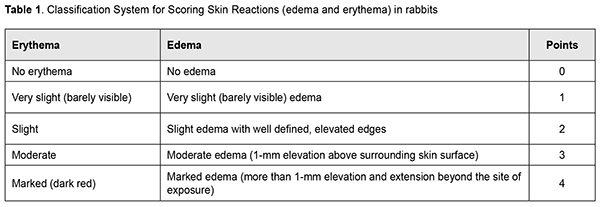
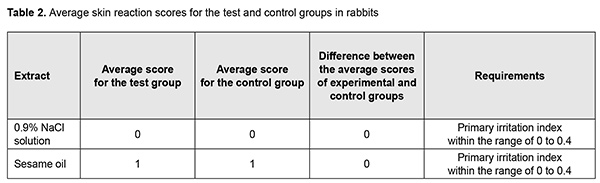
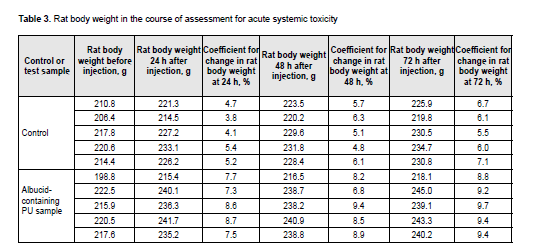
http://doi.org/10.31288/oftalmolzh202063037 Received: 23 April 2020; Published on-line: 21 December 2020 Response of soft tissues and abdominal organs of rabbits and rats to implanting albucid-containing cross-linked polyurethane composite N. A. Galatenko1, R. A. Rozhnova1, D. V. Kuliesh1, T. V. Visloguzova1, A. P. Maletskyy2, N. M. Bigun3 1 Institute for Chemistry of High-Molecular Compounds, Academy of Science of Ukraine; Kyiv (Ukraine) 2 The Filatov Institute of Eye Diseases and Tissue Therapy of the NAMS of Ukraine; Odesa (Ukraine) 3 Lviv Regional Clinical Hospital; Lviv (Ukraine) E-mail: maletskiy@filatov.com.ua TO CITE THIS ARTICLE: Galatenko NA, Rozhnova RA, Kuliesh DV, Visloguzova TV, Maletskyy AP, Bigun NM. Response of soft tissues and abdominal organs of rabbits and rats to implanting albucid-containing cross-linked polyurethane composite. J.ophthalmol.(Ukraine).2020;6:30-7. http://doi.org/10.31288/oftalmolzh202063037 Background: Craniofacial injuries represent 29% of all trauma cases, and patients need to have their affected orbit, orbital adnexa, and periorbital area surgically reconstructed or restored. However, the outcomes of these procedures depend also on the quality of implant materials. Previously, we have developed a polymer material made of cross-linked polyurethane (PU) and containing a biologically active substance, albucid; it seems to be a promising implant material. Purpose: To investigate experimentally the response of soft tissues and abdominal organs of animals (rabbits and rats) to implanting the albucid-containing cross-linked PU composite. Material and Methods: Assessment of soft tissue response to implantation of the synthetic polymer material. The skin response to implanting the albucid-containing cross-linked PU composite was assessed through the intracutaneous injection of the extract of the test material in rabbits. The soft tissue response was assessed through subcutaneous implantation of cross-linked PU composites in Wistar rats. The response of abdominal organs to implanting the cross-linked PU composite was assessed through the intraperitoneal injection (20 ml per kg body weight) of the extract of the test material in Wistar rats. Conclusion: The intracutaneous injection of the extract of the test material caused neither erythema nor edema in rabbits, and the test sample of the albucid-containing cross-linked PU composite was considered non-irritating, since a difference between the average scores for the test extract and control extract (i.e., a value of the primary irritation index) was of 0 to 0.4 points. Over the period of observation of potential acute systemic toxicity, no animal injected with the extract of the test material (the albucid-containing cross-linked PU composite) displayed higher biologic response than animals injected with the control extract, and the test sample of the albucid-containing cross-linked PU composite conformed to the requirements of tests for systemic toxicity. This study demonstrated a natural process for a foreign body residing in the body (gradual foreign body separation from the surrounding tissues due to formation of a connective tissue capsule) after implantation of the samples of cross-linked PU composites in animal bodies. The cellular responses were (1) typical for a living body response to the presence of a foreign body at the site of implant placement and (2) characteristic for aseptic inflammation. The test samples produced moderate irritation when placed into the animal’s body. Ключові слова: сітчастий поліуретан, альбуцид, імплантація, реконструктивні операції на органі зору, гостра системна токсичність, локальні ефекти
Introduction Penetrating globe injury is a leading cause of registered visual disability among working-age adults. Intraocular foreign bodies (IOFBs) account for 18%-41% of all penetrating globe injuries. The visual prognosis of an injury with an IOFB is determined by the size, composition, depth and location of the foreign body, and its duration in situ [1-3]. Endophthalmitis is one of the most severe potential complications of penetrating globe injury. Among patients with infectious endophthalmitis, post-traumatic endophthalmitis comprises about 10–30% of cases. Factors associated with increased risk of endophthalmitis following penetrating trauma include retained IOFB, delay in wound closure of >24 h and injury in a rural setting. Soil contamination is believed to result in a higher rate of endophthalmitis for open globe injuries occurring in a rural versus a non-rural setting. Endophthalmitis developed in 30% of patients with rural penetrating trauma, compared with 11% of patients with non-rural penetrating trauma [4, 5]. The incidence of endophthalmitis was higher after IOFB-related penetrating globe injury than after penetrating globe injury without IOFB [6]. Metallosis is a dangerous complication after metallic IOFB-related penetrating globe injury. It occurs through damage to intraocular tissue through toxic effects of retained metallic IOFB, these effects manifesting clinically as recurrent episodes of choroidal inflammation. Clinical manifestations developing as early as the first weeks after the traumatic event and cases of metallic foreign bodies presenting years after the initial trauma without causing severe ocular inflammatory response have been rarely reported [7-11]. Therefore, early detection of an IOFB is not only important for selecting the most appropriate surgical treatment strategy but also a factor influencing the treatment outcome. Despite continuous advances in imaging techniques, early detection of an IOFB is still a challenge for the ophthalmologist. Visualizing a ciliary body IOFB is especially challenging. Approximately 5% of IOFBs are found in the projection of or close to the ciliary body [3, 7]. Computed tomography (CT), radiography with the Komberg-Baltin prosthesis, ultrasonography and magnetic resonance imaging (MRI) are the most effective techniques for detecting and localizing an IOFB. Nevertheless, there are an ample number of reports of missed anterior chamber IOFBs [8, 10]. We have previously demonstrated that near-infrared transpalpebral transillumination (NIR TPT) is a modality enabling easy and non-invasive visualization and assessment of the sizes of ciliary body structures [11-13]. In addition, NIR TPT enables visualization of shadows of ciliary body IOFBs of various origin (metallic, stone and wood) in patients with penetrating globe injuries. The use of NIR TPT as means for visualizing an IOFB in conjunction with traditional imaging modalities seems promising and would improve our ability to diagnose these patients preoperatively [14-16]. The purpose of the study was to compare detection rates of near-infrared transpalpebral transillumination, ultrasonography and radiography for foreign bodies situated in the anterior segment of the eye. Material and Methods This was an open-label, prospective and non-interventional study. This study was performed within the framework of a planned research design and the study protocol was approved by the Bioethics Committee of the Filatov Institute. The study followed the ethical standards stated in the Declaration of Helsinki, the European Convention on Human Rights and Biomedicine and relevant laws of Ukraine. Informed consent was obtained from all study subjects. Thirty male patients (30 eyes; age, 21 to 65 years) with penetrating globe injuries and suspected foreign body in the anterior segment of the eye (the anterior chamber, lens, anterior vitreous cavity) were under our observation. They underwent visual acuity assessment, biomicroscopy, ophthalmoscopy, metal detector examination, anteroposterior and lateral radiography (both with and without using the Komberg-Baltin prosthesis), ultrasound biometry, ultrasound scanning of the anterior eye and posterior eye; ultrasound distant perimetry; metal detector examination; and NIR TPT. A diagnosis of IOFB was confirmed or denied by the three imaging modalities (NIR TPT, ultrasound scanning and X-ray examination) using the signs that were specific for each of the modalities. The NIR TPT system used consisted of (1) a wireless infrared light-emitting-diode light source with a dominant wavelength of 940 nm and (2) slit-lamp attachable monochrome video camera (Blackfly®, FLIR Integrated Imaging Solutions Inc., Canada) capable of recording NIR images and video [14, 15, 17]. The transpalpebral illumination study was performed with the patient sitting at the slit lamp. Images of scleral shadows of the IOFB, pars plicata and pars plana were taken and saved in the computer. We used the Aviso ultrasound (Quantel Medical; Cournon d'Auvergne, France) with 10-MHz and 50-MHz linear probes for the posterior and anterior segments of the eye, respectively. The ultrasound study was performed with the patient lying on his back, and with the head of the bed raised. In the ultrasound study with the anterior probe, patients were administered topical anesthesias with ophthalmic 0.5% proparacaine hydrochloride (ALCAINE®, SA Alcon-Couvreur NV, Puurs, Belgium), and, subsequently, Vidisic Eye Gel (Dr. GERHARD MANN Chem.-Pharm. Fabrik, Berlin, Germany) as a contact gel. The posterior probe was placed over the patient’s closed eyelid to scan the eye non-invasively. Targeted anteroposterior and lateral radiography (with the Komberg-Baltin prosthesis) was performed using a Proteus XR/a radiography system (GE Healthcare, Milwaukee, Wis), and patients were administered topical anesthesias with ophthalmic 0.5% proparacaine hydrochloride (ALCAINE®). Contact invasive imaging studies (radiography with the Komberg-Baltin prosthesis, ultrasound scanning of the anterior eye and ultrasound biometry) were not performed in patients with a non-adapted corneal or scleral wound. Receiver operating characteristic (ROC) curves were constructed to obtain graphical representation of selectivity and specificity, and the area under the ROC curve was calculated to compare the efficacy of various imaging techniques for detection of foreign bodies in the anterior eye. Predictive characteristics for the techniques for the detection of IOFBs are reported as mean and 95% confidence intervals (CI). Statistical analyses were conducted using Statistica 10.0 (StatSoft, Tulsa, OK, USA) software [18]. Results An intraocular foreign body was detected in the anterior segment (the anterior chamber, lens, anterior vitreous cavity) in all the 30 patients. Of the 30 eyes with IOFB, a foreign body was identified and localized by radiography in 21 eyes (70%). Radiography failed to detect a foreign body in the rest 9 eyes (30%) because the foreign body was either composed of organic material (rock or wood) or metallic and small (less than 1.0 mm). Of note that, in 6 patients (20%), a foreign body was found by radiography, but it was not possible to localize it with the Komberg-Baltin prosthesis due to the non-adapted wound of the globe and associated risk of iatrogenic complications. Radiography showed no false positives, but 3 false negatives, all of which were associated with small (≤0.5 x 0.5 mm) metallic foreign bodies. Of the 30 eyes with IOFB, a foreign body was identified by ultrasound in 24 eyes (80%). Ultrasound scanning of the anterior eye could not be performed in 6 patients (20%) due to the non-adapted wound of the globe. Ultrasonography showed no false positives. NIR TPT revealed a foreign body in the anterior eye in 28 eyes (93.3%) of the 30 eyes with IOFB. Particularly, NIR TPT found a foreign body in the anterior segment in 3 eyes in which either an IOFB was missed by radiography and ultrasound, or ultrasound could not be performed. NIR TPT failed to detect a foreign body in 2 eyes (6.6%). In one of these two eyes, a foreign body could not be detected by NIR TPT because of shielding effect of massive subconjunctival hemorrhage, but it was detected both by radiography and ultrasound. In the other eye, neither of the imaging modalities could detect the IOFB because the latter was too small, but the foreign body was revealed during surgery. In addition, two eyes were mistakenly found to have a foreign body in the anterior vitreous (false positive results) by NIR TPT, but this was subsequently confirmed neither by radiography nor by ultrasound. These two patients had subconjunctival and vitreous hemorrhages. Of note is that the method we have developed (1) enabled the diagnosis of IOFB in swollen and opaque lens masses and opaque corneas, and (2) correctly detected not only X-ray positive foreign bodies, but also X-ray-negative foreign bodies. ROC analysis of the IOFB detection methods was performed for 50 eyes; the presence of a foreign body was confirmed for 30, and the absence of a foreign body was confirmed for 20 of these eyes. ROC curves for the three IOFB detection methods are shown in Figure 1. The area under curve (AUC) for NIR TPT was 0.92 (95% CI, 0.80–0.98); for radiography, 0.82 (95% CI, 0.68–0.91), and for ultrasound, 0.87 (95% CI, 0.74–0.95), demonstrating good efficacy of all the three IOFB detection methods, although there was no significant difference (p > 0.05) in AUC between each two of them. Table 2 shows predictive characteristics of the methods under analysis.
Discussion Inadequate foreign body visualization is the main reason of unsuccessful removal attempts and late removal of foreign bodies situated in the anterior segment of the eye [19-21]. Although radiography is an effective imaging modality enabling detection and accurate localization of an IOFB, detecting an X-ray negative foreign body by radiography is difficult. Thus, the foreign bodies composed of organic materials (stone or wood) were missed by radiography in our study. In addition, small (≤ 1.0 mm) metallic foreign bodies may be missed by radiography due to their shape and/or location in the anterior chamber angle, beneath the iris, or in the ciliary body area, which was confirmed by the current study. Moreover, accurate IOFB localization by radiography with the Komberg-Baltin prosthesis sometimes cannot be performed due to the non-adapted wound of the globe and/or associated risk of iatrogenic complications. Ultrasonography is another imaging modality proved to be effective in detecting foreign bodies of various sizes, shapes, materials and locations (including the anterior segment of the eye). In some cases, however, ultrasonography should not be performed due to high risk of iatrogenic complications. Patients with non-adapted corneal or scleral defects may have additional wound infection; prolapse of the retina, choroid or sclera; or iatrogenic expulsive hemorrhage due to globe contact with or compression by the probe that is applied directly upon the surface of the globe [22]. Thus, ultrasound study of the anterior segment of the eye could not be safely performed in 19.8% of study patients due to the presence of a non-adapted globe wound, which limited out ability to diagnose IOFB in these patients. The use of near-infrared LED illumination in NIR TPT enables transillumination not only through the sclera, but even through the patient’s lid. In addition, absence of physical contact between LED light source and the cornea and sclera allows excluding complications and trauma of these ocular structures. We managed to conduct the NIR TPT procedure quickly and safely with as little pain and discomfort to the patient as possible in all cases of the study. Particularly, no iatrogenic damage was observed after performing the procedure in patients with non-adapted corneal and scleral defects. The use of LED as a source of infrared light simplifies the globe transillumination system, and there is no need for the use of optic fibers to transmit the light radiation to the eye. The LED light source is a compact wireless device that needs no additional infrared filters. One may use infrared LEDs of different wavelengths to achieve better visualization of the globe. With the use of near-infrared light for globe transillumination, there is no glare from bright visible light, which makes the conduct of studies in this patient category easier. The proposed visualization approach enables taking real-time photographs and shooting real-time video of TPT pictures [11-13]. In addition, we managed to visualize IOFBs of various compositions and dimensions, particularly, in the presence of opaque media using NIR TPT. Thus, NIR TPT found a foreign body in the anterior eye in the three eyes (10% of the study patients) in which either an IOFB was missed by radiography and ultrasound, or ultrasound could not be performed due to the presence of a non-adapted globe wound. Moreover, NIR TPT enables visualizing ciliary body structures and localizing and measuring the dimensions of shadows cast by these structures on the sclera [14-16]. Thus, in the current study, we managed to localize an IOFB shadow with respect to shadows cast by ciliary body structures on the sclera, which allowed localization of a foreign body and selecting the most appropriate surgical treatment strategy. The method of NIR TPT, however, has certain drawbacks. For example, a foreign body in the projection of the ciliary body is difficult to identify in the presence of massive subconjunctival hemorrhage due to a shielding effect of blood with respect to the near-infrared light. In addition, intensive absorption of near-infrared light by subconjunctival blood in some cases led to incorrect assessment of NIR TPT pictures and false positive results. The use of a non-invasive NIR TPT in conjunction with traditional imaging modalities (radiography and ultrasound) resulted in a ten percent increase in the rate of detection of anterior segment foreign bodies due to the detection of some X-ray-negative IOFBs and identification of small (less than 1 mm) IOFBs.
References 1.Grusha OV, Grusha IaO. [Five Hundred Orbital Plastic Repairs: Analysis of Complications]. Vestn Oftalmol. 2006;1:22-4. Russian. 2.Gundorova RA, Neroiev VV, Kashnikov VV. [Eye injuries]. Moscow: GEOTAR-Media; 2009. Russian. 3.Lowery J, Carlson AN, Abelson MB, et al. 2001 year in review. Rev Ophthalmol. 2001; 8(11):73-87. 4.Grishina NI, Fedorischeva LE, Kolbenev IO. Traumatic orbital injuries, the methods of diagnosis and treatment. In: Brovkina A, editor. Collection of research papers “Diseases, tumors and traumatic injuries of the orbit”. Moscow; 2005. p. 87–90. 5.Astakhov YuS, Nikolaenko VP, D’yakov VE. [Use of polytetrafluoroethylene implants in ophthalmic surgery]. St Petersburg: Foliant; 2007. Russian. 6.Galatenko NA, Kuliesh DV, Maletskyi AP, Karpenko OS. Soft-tissue response to synthetic polymer implants made of cross-linked polyurethane and containing a biologically active substance, albucid or dacarbazine, in animals. J Ophthalmol (Ukraine). 2018;6:52-8. 7.Karaian AS. [One-stage repair of traumatic defects and deformations of the chhekbone, nose, and orbit complex]. [Dr Sc (Med) Dissertation]. Moscow: Central Research Institute for Dentistry and Maxillofacial Surgery; 2008. Russian. 8.Krasilnikova VL. [Foamed ceramics- and hydroxyapatite-based ocomotor stump of the ocular prosthesis (experimental study)]. [Cand Sc (Med) Thesis]. St Petersburg: Russian Medical Academy of Postgraduate Education; 2002. Russian. 9.Maletskyy AP, Zubok DI. [Possible surgical approaches to the treatment of trauma and tumors of the eyelids, orbit and oculoorbital area]. In: Proceedings of the Ophthalmology Conference on the Occasion of the 80th Anniversary of the Ukrainian Society of Ophthalmologists. September 12-13, 2018, Vinnytsia. P. 11-3. Russian. 10.Hintschich C. [Dermis-fat graft. Possibilities and limitations]. Ophthalmologe. 2003 Jul;100(7):518-24.. 11.Galatenko NA, Rozhnova RA. [Biologically active polymeric materials for medicine]. Kyiv: Naukova Dumka; 2013. Russian. 12.Rozhnova RA, Shilov VV, Galatenko NA. [Structural and morphological studies of biologically active implants with prolonged healing action]. Kompozitsiini polymerni materialy. 2000;22 (2): 146-50. Russian. 13.Lipatova TE, Pkhakadze GA. [Polymers in endoprosthetics]. Kyiv: Naukova Dumka; 1983. Russian. 14.European convention for the protection of vertebrate animals used for experimental and other scientific purposes. Council of Europe, Strasbourg; 1986. 15.Maletskyy AP, Dubkova VI, Maievskaia OI, Bigun NM. [Results of the use of polymer composite Ikvoban in reconstructive surgery of the orbit and oculo-orbital region]. Oftalmologiia. Vostochnaia Evropa. 2013;special issue:188-97. Russian. 16.Sarkisov DS, Petrova YuL. [Microscopic technique]. Moscow: Meditsina; 1996. Russian. The authors certify that they have no conflicts of interest in the subject matter or materials discussed in this manuscript.
|