J.ophthalmol.(Ukraine).2019;5:18-21.
|
http://doi.org/10.31288/oftalmolzh201951821 Received: 15 July 2019; Published on-line: 30 October 2019 Blood sICAM-1 levels in type 2 diabetes mellitus patients with various grades of DME S.A. Suk1, Cand Sc (Med); M. L. Kyryliuk2, Dr Sc (Med), Prof.; S. O. Rykov1, Dr Sc (Med), Prof. 1 Shupik National Medical Academy of Postgraduate Education, Ministry of Health of Ukraine; Kyiv (Ukraine) 2 Ukrainian Research Center for Endocrine Surgery, Transplantation of Endocrine Organs and Tissues; Kyiv (Ukraine) E-mail: sasuk1972@gmail.com, mlkyryliuk@endosurg.com.ua, eye-bolit@ukr.net TO CITE THIS ARTICLE: Suk SA, Kyryliuk ML, Rykov SO. Blood sICAM-1 levels in type 2 diabetes mellitus patients with various grades of DME. J.ophthalmol.(Ukraine).2019;5:18-21. http://doi.org/10.31288/oftalmolzh201951821
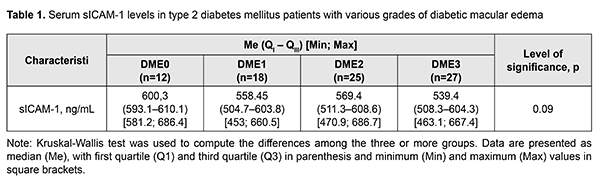
Background: Diabetic macular edema (DME) is a major cause of visual impairment in type 2 diabetes mellitus (DM) patients. Non-specific inflammation is an important factor of the underlying processes of DME. Intercellular adhesion molecules (ICAM), particularly, soluble ICAM-1 (s-ICAM-1), are a local inflammatory mediator involved in the pathogenesis of diabetic injury to the layers of the eye. The literature is scant on the assessment of sICAM-1 blood levels in type 2 DM patients with diabetic injury to the neurovascular system of the eye. Purpose: To investigate serum sICAM-1 levels in type 2 DM patients with various grades of DME. Materials and Methods: Eighty-two type 2 DM patients (145 eyes) were involved in the study; they were divided into four groups based on DME severity. Patients underwent laboratory examination (glycated hemoglobin A1c, HbA1c) and comprehensive eye examination. Inclusion criteria were voluntary informed consent, age above 18 years, and presence of type 2 DM. Exclusion criteria were pregnancy, presence of endocrine disorders that may lead to type 2 DM (Cushing's syndrome, acromegalia, dispituitarism, polycystic ovaries syndrome, etc.), coagulation system impairment, neurodegenerative disorders of the central nervous system, type 1 DM, acute infectious disorders, cancer, decompensation of concomitant medical conditions, mental disorders, administration of neuroleptics and/or antidepressants, or presence of proteinuria, glaucoma or mature cataract. Serum sICAM-1 levels were determined by enzyme-linked immunosorbent assay (ELISA). Single-factor analysis of variance was used for statistical analysis. Results: The median serum sICAM-1 level value for DME0 group (Me = 600.3 ng/mL (593.1 ng/mL – 610.1 ng/mL)) was statistically significantly (p = 0.01) higher than for conventionally amalgamated (DME1 – DME3) group (Me = 558.5 ng/mL (506.9 ng/mL – 607 ng/mL)). Conclusion: The median serum sICAM-1 level value for patients with DME0 (Me = 600.3 ng/mL (593.1 ng/mL – 610.1 ng/mL)) was statistically significantly (p = 0.01) higher than for those with DME1 to DME3 (Me = 558.5 ng/mL (506.9 ng/mL – 607 ng/mL)). Keywords: diabetic macular edema, type 2 diabetes mellitus, sICAM-1 Introduction Diabetic macular edema (DME) is a major cause of visual impairment in diabetic patients as type 2 diabetes mellitus (DM) has a high prevalence worldwide. The available data on the principal pathogenetic components of DME evidence that non-specific inflammation is an important factor of the underlying processes of DME [1]. Intercellular adhesion molecules (ICAM), particularly, soluble ICAM-1 (s-ICAM-1), are a local inflammatory mediator involved in the pathogenesis of diabetic injury to the retina. ICAM-1 molecules are available at low concentrations on the membranes of white cells and endothelial cells. ICAM-1 expression on the cytoplasmic membrane abruptly increases after stimulation with cytokines, especially interleukin-1 (IL-1) and tumor necrosis factor (TNF). ICAM-1 is a ligand of lymphocyte function-associated antigen-1 (LFA-1) found on the white blood cells which after recruiting are activated, become bound to the endothelium due to the ICAM-1/LFA-1 complex, and penetrate to the site of inflammation [2]. Thus, the white cell adhesion mediated by ICAM-1 and CD18 increases in the retina of diabetic mice or under conditions of experimental hypergalactosemia and may explain many of important blood-retinal barrier injuries in diabetic retinopathy [3]. Unfortunately, the literature is scant on the assessment of sICAM-1 blood levels in type 2 DM patients with diabetic injury to the neurovascular system of the eye [4]. Purpose: To investigate serum sICAM-1 levels in type 2 DM patients with various grades of diabetic macular edema. Materials and Methods Eighty-two type 2 DM patients (145 eyes; mean age, 65.25±10.85 years; mean diabetes duration, 14.0±7.05 years; mean HbA1с, 8.40±1.58%) were involved in the study; they were divided into four groups based on DME severity. Patients underwent laboratory examination (glycated hemoglobin A1c, HbA1c) and comprehensive eye examination. They were divided into four groups based on DME severity as per the International Diabetic Macular Clinical Edema Severity Scale [5] (DME0, DME apparently absent; DME1, mild DME; DME2, moderate DME; DME3, severe DME). Inclusion criteria were voluntary informed consent, age above 18 years, and presence of type 2 DM. Exclusion criteria were pregnancy, presence of endocrine disorders that may lead to type 2 DM (Cushing's syndrome, acromegalia, dispituitarism, polycystic ovaries syndrome, etc.), coagulation system impairment, neurodegenerative disorders of the central nervous system, type 1 DM, acute infectious disorders, cancer, decompensation of concomitant medical conditions, mental disorders, administration of neuroleptics and/or antidepressants, or presence of proteinuria, glaucoma or mature cataract. This study was performed in full compliance with the treatment and examination guidelines provided in the Helsinki declaration (as revised in 2008) and relevant orders issued by the Ministry of Health of Ukraine (No. 281 issued 01.11.2000, No. 355 issued 25.09.2002, No. 356 issued 22.05.2009 (as revised in Order No. 574 issued 05.08.2009), and No. 1118 issued 21.12.2012). HbA1c levels were measured by high-performance liquid chromatography, with Bio-Rad D10 analyzer (Bio-Rad Laboratories, CA) and assay. Serum sICAM-1 levels were measured by enzyme-linked immunosorbent assay (ELISA) with IEMS Reader MF analyzer (Labsystems, Helsinki, Finland) and Human sICAM-1 Platinum ELISA Kit, Extra Sensitive (Bender MedSystem GmbH, Austria). The eye examination included measurements of visual acuity, perimetry, tonometry, refractometry, biomicroscopy, gonioscopy, ophthalmoscopy, optical coherence tomography with angiography, and fundoscopy (with fundus photography). Statistical analyses were performed using MedCalc v 18.11 software (MedCalc Software Inc, Broekstraat, Belgium). The Shapiro–Wilk test was used to control quantitative data for normality. Single-factor analysis of variance (Kruskal-Wallis test) was used to compute the differences among the three or more groups. Data are presented as median (Me) (with first quartile (Q1) and third quartile (Q3) in parenthesis) and minimum and maximum values. The level of significance p ? 0.05 was assumed [6]. Results Table 1 presents serum sICAM-1 levels in type 2 DM patients with various grades of diabetic macular edema.
There were no significant differences among groups of type 2 DM patients with various grades of DME in serum sICAM-1 levels (p = 0.09). However, the median serum sICAM-1 level value for DME0 group (Me = 600.3 ng/mL (593.1 ng/mL – 610.1 ng/mL)) was statistically significantly higher than for conventionally amalgamated (DME1 – DME3) group (Me = 558.5 ng/mL (506.9 ng/mL – 607 ng/mL)). Discussion The median serum sICAM-1 levels for our type 2 DM patients with DME were 2.34 to 2.61 times higher than the reference levels for healthy donors specified in kit instructions. In addition, the median serum sICAM-1 level value for conventionally amalgamated (DME1 – DME3) group was statistically significantly lower than for DME0 group. It has been reported [7] than vitreous levels of both VEGF and sICAM-1 were significantly higher in patients with hyperfluorescent DME than in those with minimally fluorescent DME (P = 0.0027 and P = 0.0005, respectively). The vitreous levels of both VEGF and sICAM-1 were significantly correlated with retinal thickness at the central fovea (P < 0.0001 and P = 0.0005, respectively). These results suggested that VEGF and ICAM-1 are related to the increase of vascular permeability in DME patients [6]. In the study by Zhu et al [8], aqueous sICAM-1, VEGF and MCP-1 levels in DME group were statistically significantly higher than in controls [11]. Hiller et al [9] investigated aqueous humor cytokine level changes in response to intravitreal ranibizumab therapy for the management of DME. Aqueous ICAM-1 levels were decreased compared to baseline as early as month 2, and continued decreasing at month 3 [9]. In general, type 2 DM impairs the balance between neurotropic factors contributing to cell survival (neurotrophins) and proinflammatory mediators, leading to hyperactive inflammatory responses in the retinal endothelium, micro- and microglia, and emergence of inflammatory mediators, which causes increased vascular permeability, arrest of capillary blood flow ICAM-1, VCAM-1, PAI-1) [10, 11], apoptosis of endothelial cells, macular edema and neovascularization (VEGF). Conclusion The median serum sICAM-1 level value for DME0 group (Me = 600.3 ng/mL (593.1 ng/mL – 610.1 ng/mL)) was statistically significantly higher than for conventionally amalgamated (DME1 – DME3) group (Me = 558.5 ng/mL (506.9 ng/mL – 607 ng/mL)). References 1.Pasyechnikiva NV, Suk SA, Kuznetsova TA, Parkhomenko OG. [Diabetic maculopathy. Current aspects of the pathogenesis, clinical picture, diagnosis and treatment]. Kyiv: Karbon LTD;2010. Russian. 2.Yang L, Froio RM, Sciuto TE, et al. ICAM-1 regulates neutrophil adhesion and transcellular migration of TNF-alpha-activated vascular endothelium under flow. Blood. 2005 Jul 15;106(2):584-92. 3.Abu El-Asrar, AM, Al-Mezaine HS, Ola MS. Pathophysiology and Management of Diabetic Retinopathy. Expert Rev Ophthalmol. 2009;4(6):627-47. 4.Xie Z, Liang H. Association between diabetic retinopathy in type 2 diabetes and the ICAM-1 rs5498 polymorphism: a meta-analysis of case-control studies. BMC Ophthalmology. 2018;18:297. 5.American Academy of Ophthalmology Retina -Vitreous Panel. Preferred Practice Pattern® Guidelines. Diabetic Retinopathy. San Francisco, CA: American Academy of Ophthalmology; 2014. Available at: www.aao.org/ppp. 6.Jekel JF, Katz DL, Elmore JG, Wild DMG. Epidemiology, biostatistics, and preventive medicine. 3rd ed. Philadelphia: Saunders Elsevier; 2007. 7.Funatsu H., Yamashita H., Sakata K. et al. Vitreous Levels of Vascular Endothelial Growth Factor and Intercellular Adhesion Molecule 1 Are Related to Diabetic Macular Edema. Ophthalmology. June 2005; 112(5):806-16. DOI: 10.1016/j.ophtha.2004.11.045 8.Zhu D, Zhu H, Wang C, Yang D. Intraocular soluble intracellular adhesion molecule-1 correlates with subretinal fluid height of diabetic macular edema. Indian J Ophthalmol. 2014 Mar;62(3):295-8. doi: 10.4103/0301-4738.111184. 9.Hillier RJ, Ojaimi E, Wong DT, et al. Aqueous Humor Cytokine Levels and Anatomic Response to Intravitreal Ranibizumab in Diabetic Macular Edema. JAMA Ophthalmol. 2018 Apr 1;136(4):382-388. 10.Pylypenko LYu, Serdiuk VM. [Effect of diabetic retinopathy progression factors on the content of marker of adhesive endothelial dysfunction VCAM-1 in the blood at type 2 diabetes mellitus as a cluster of metabolic syndrome]. Archive of Ukrainian Ophthalmology. 2018;6(1(10)):42-7. Russian. 11.Serdiuk VM, Kyryliuk ML, Pylypenko LYu. Activity of plasminogen activator inhibitor-1 in blood of patients with metabolic syndrome depending on a stage of diabetic retinopathy. J Ophthalmol (Ukraine). 2018;3:52-56.
All authors declare that they have neither financial nor non-financial competing interests.
|