J.ophthalmol.(Ukraine).2017;2:44-50.
|
https://doi.org/10.31288/oftalmolzh201724450 Current antibacterial therapy for ocular burns
S.A. Iakymenko, Dr. Sc. (Med), Prof. E.A. Khrustaliova O.I. Buznyk, Cand. Sc. (Med), MD P.O. Kostenko, Cand. Sc. (Med) A.L. Molodaia, L.V. Dolenko Filatov Institute of Eye Diseases and Tissue Therapy Odessa, Ukraine
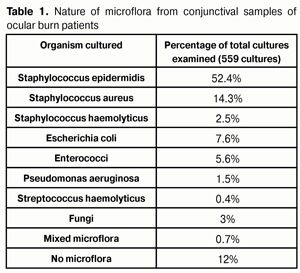
Background: Since ocular infection is common following ocular burns, and may lead to severe infectious complications like corneal ulcers, infiltrates and abscesses, prevention and treatment of these complications has been always important. Purpose: To investigate the nature of the microflora cultured from ocular burn patients and the sensitivity of this microflora to current antibacterial agents. Materials and Methods: We investigated conjunctival microflora from 184 ocular burn patients (224 eyes) at different time points following ocular burns. In total, 559 cultures were performed. Antibacterial and antiseptic agents under investigation included ciprofloxacin (Floximed), ofloxacin, levofloxacin (Levofloximed), moxifloxacin, gentamycin, tobramycin (Tobrimed), futaron, octenisept and betadine. Results: Microflora was cultured from 88% of the conjunctival samples examined. Staphylococcus epidermidis was the most commonly cultured organism (54.2%), followed by Staphylococcus aureus (14.3%), Escherichia coli (7.6%), Enterococci (5.6%), fungi (3.0%), Staphylococcus haemolyticus (2.5%), Pseudomonas aeruginosa (1.5%), and Streptococcus haemolyticus (0.4%). No organisms were isolated from 12% of the conjunctival microflora samples. The sensitivity of these microorganisms to each of the antibacterial agents under investigation was determined. Levofloxacin (Levofloximed), ciprofloxacin (Floximed), moxifloxacin, ofloxacin, gentamycin, and tobramycin (Tobrimed) demonstrated high antibacterial activity against the organisms. Conclusion: Following ocular burns, conjunctival microflora always contains organisms that can cause various infectious complications. Administration of antibacterial medications for prevention and treatment of infectious complications should be based on the sensitivity of the organisms to these medications. Prior to obtaining the results of sensitivity analyses of cultured microorganisms, or if such tests are unavailable, broad spectrum antibiotics, ? fluoroquinolones (moxifloxacin, levofloxacin (Levofloximed), and ciprofloxacin (Floximed)) and aminoglycosides (gentamycin and tobramycin (Tobrimed)) ? should be administered. If the microorganism exhibits no sensitivity to antibiotics, such antiseptics as octenisept or betadine may be administered. Key words: ocular burns, microflora, antibacterial therapy Introduction Necrotic tissue provides a medium for the growth of pathogenic microbes that can cause severe infectious complications (corneal ulcers, infiltrates and abscesses) following burns, and previously we have found evidence of ocular infection in 84% of the eyes following ocular burn injuries. In addition, necrotic tissue releases proteases that contribute to breakdown of the tissue damaged by burn. Therefore, prevention and treatment of infectious complications following ocular burns have been always important [1, 2]. On the other hand, since widespread use of antibiotics resulted in a rise of resistant organisms, a regime of antibiotic treatment must be based on knowledge of the antibiotic sensitivity patterns of the cultured microflora [1, 3-5]. The purpose of the study was to investigate both the nature of the microflora cultured in ocular burns and the sensitivity of this microflora to the antibacterial agents most commonly used today by practitioners (including us) in the treatment and prevention of infectious complications following ocular burns. Materials and Methods We investigated conjunctival microflora from 184 ocular burn patients (224 eyes; 104 males and 80 females; age, 18 years to 72 years) of the Ocular Burn Injury Department at the Filatov Institute at different times following burn injury. In total, 559 cultures were performed for these patients. Samples were obtained from the conjunctival sac, cultured on Muller-Hinton agar medium, and the sensitivity pattern to the antibacterial agent under study was determined at the Bacteriologic laboratory of the Institute using a standard methodology [6, 7]. Antibacterial and antiseptic agents under investigation included ciprofloxacin (Floximed), ofloxacin, levofloxacin (Levofloximed), moxifloxacin, gentamycin, tobramycin (Tobrimed), futaron, octenisept and betadine. Results and Discussion Microflora was cultured from 88% of the conjunctival samples examined. Staphylococcus epidermidis was the most commonly cultured organism (54.2%), followed by Staphylococcus aureus (14.3%), Escherichia coli (7.6%), Enterococci (5.6%), fungi (3.0%), Staphylococcus haemolyticus (2.5%), Pseudomonas aeruginosa (1.5%), and Streptococcus haemolyticus (0.4%). No organisms were isolated from 12% of the conjunctival microflora samples. Two or more of the organisms (Table 1) were cultured from 0.7% of samples. No substantial changes in the nature of the conjunctival microflora were identified at different treatment time points after admittance to the clinic. The incidence of detection of S. aureus and Escherichia coli in the conjunctival microflora somewhat increased during the treatment process.
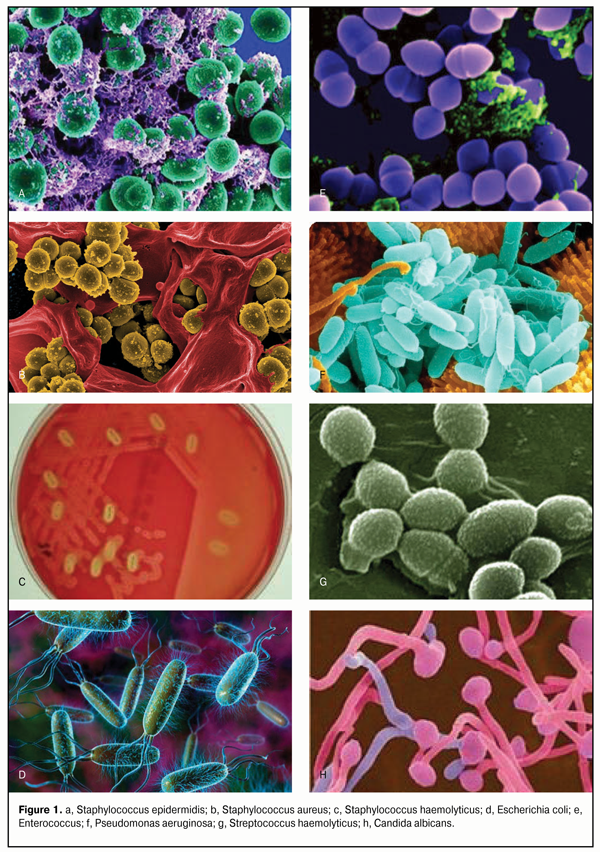
Staphylococci are Gram-positive, non-motile, spherical bacteria. The genus Staphylococcus has numerous species (at least 27), of which S. aureus, S. epidermidis and S. haemolyticus are the most common species. Staphylococci are opportunistic bacteria, i.e., those that can only cause symptomatic disease under optimal conditions. The danger these organisms cause is that they have ability to produce toxins and enzymes injurious to vital cellular activities. Staphylococcus epidermidis (Fig. 1a) belongs to the group of coagulase-negative staphylococci. These bacteria are cocci about 0.5 – 1.0 ?m in diameter, and occur usually in irregular grapelike clusters. Staphylococcus epidermidis was cultured from 52% of the conjunctival samples examined. The most common locations of human colonization of the organism are the skin and mucous membranes. Staphylococcus epidermidis is generally considered a low-virulence organism and is often encountered in patients with low resistance, but it also may cause disease when conditions allow. Therefore, if burns occur, attempts should be made to eliminate this pathogen.
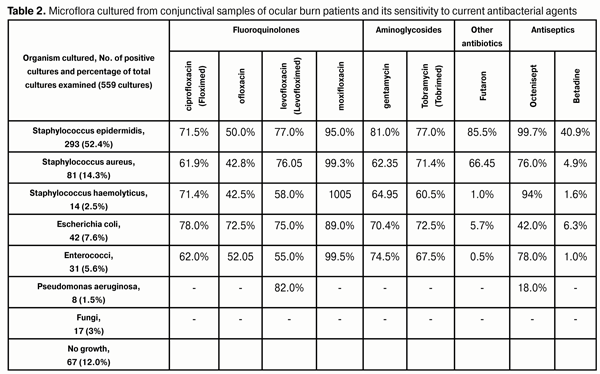
Staphylococcus aureus (Fig. 1 b) is an extremely adaptable and versatile organism that is implicated in practically any problem related to stafilococci, and expresses a variety of virulence factors. The organism was cultured from 14.3% of the conjunctival samples examined. These bacteria were given the specific epithet Staphylococcus aureus because of the yellow-to-orange microscopic colony appearance. The organism is markedly resistant to external environmental conditions. S. aureus capsule induces inflammatory and allergic responses, while neutralizing immunoglobulins immobilize phagocytes. These pathogens secrete numerous enzymes that degrade cell walls and reduce the effect of antibiotics. In addition, special enzyme, coagulase, is secreted that targets the blood clotting cascade, and bacteria become entrapped within microthrombi. Moreover, S. aureus produces hemolysins that destroy red blood cells, white blood cells, etc, and releases at least ten toxins with different modes of action. Staphylococcus haemolyticus (Fig.1c), an opportunistic pathogen, colonizes the skin and mucous membranes of humans. The organism was cultured from 2.5% of the conjunctival samples examined. Both the invasive and toxigenic features of the pathogen play a role in S. haemolyticus infections, and it also can cause severe infectious complications following burn injuries. Escherichia coli (Fig.1d) is a Gram negative rod (bacillus) species and a facultative anaerobe. It is a member of the normal microflora of the human intestinal tract and is regarded as an opportunistic pathogen that may cause a disease in a host that is physically impaired or debilitated. Under certain conditions, it can enhance its pathogenic potential and cause severe infectious complications. Enterococcus (Fig. 1e) is a genus of ubiquitous, Gram-positive, non-spore-forming, facultative anaerobic cocci that often occur in pairs (diplococci) or short chains and are tolerant of a wide range of environmental conditions. The organism was cultured from 5.6% of the conjunctival samples examined. These cocci are highly resistant to various disinfectants, and, over the past three decades, have acquired resistance to practically all existing classes of antimicrobial agents. High levels of endemic antibiotic resistance are the most important feature of the Enterococcus genus. Over the past two decades, especially virulent, vancomycent-resistant enterococci strains emerged that can cause nosocomial infections [6]. Pseudomonas aeruginosae (Fig.1f), the most dangerous representative of the pathogenic flora, is a species of Gram-negative, motile, rod shaped bacteria. The organism was cultured from 1.5% of the conjunctival samples examined. The pathogenic features of the organism are associated with the synthesis of various exotoxins, endotoxins and proteolytic enzymes (neutral protease, elastase and collagenase). These features become prominent most commonly in immunocompromised individuals, burn patients, and those undergoing long-term antibacterial therapy. Motility, toxins and hydrolytic enzymes contribute to the pathogenicity of Pseudomonas aeruginosae. It produces an extra-cellular glycocalyx (slime) and may occur in the environment as a glycocalyx-enclosed micro¬colony. The ability to develop and acquire antibiotic resistance contributes to the success of the pathogen. 5% chloramine, 3% hydrogen peroxide and 2% phenol are the only means effective in combating Pseudomonas aeruginosa infection. Streptococcus haemolyticus (Fig.1 g) is a Gram-negative, non-spore-forming species with marked hemolytic activity and coccoid morphology. It is ubiquitous and often colonizes the skin and mucous membranes of humans. The pathogenesis is associated with toxin production. The yeastlike fungus Candida albicans (Fig. 1h) was cultured from 3.0% of the conjunctival samples examined, more commonly from burn patients who received intensive antimicrobial therapy. Candida albicans grows as oval-shaped budding yeast (blastospore) forming pseudomycelia in cultures, tissues and exudates. Although this fungus is a component of the normal microflora of mucous membranes, it can lead to the development of a number of pathological conditions (corneal ulcers, infiltrates and abscesses), especially in frail or debilitated individuals or those on immuno-suppressive and antibacterial therapy. To prevent infectious complications following ocular burns, antibacterial treatment was prescribed on admission to the clinic, with the regime based on the antibacterial sensitivity patterns of cultured organisms. Antibacterial agents were administered as eye drops or eye spray. However, in severe ocular burns, they were administered also as peribulbar or subconjunctival injections. In addition, in some severe burn cases, intramuscular injections were administered to achieve maximum concentration of an agent in the ocular tissues. In some patients, conjunctival cultures remained positive for organisms in spite of antibacterial therapy, with the species pattern varying with time. These facts indicate that, within the treatment process following ocular burns, conjunctival microflora cultures should be taken on a regular basis (at least 1-2 times a week) for culture-directed antibacterial therapy [4]. This approach allowed us to avoid severe infectious complications in most of cases. The duration of antibacterial therapy depended on the severity of burn injury, and on whether corneal ulcers or infectious complications were present. Current antibacterial agents (fluoroquinolones, such as ciprofloxacin, ofloxacin, levofloxacin and moxifloxacin; aminoglycosides, such as gentamycin and tobramycin; and fusidic acid (futaron)) were administered to suppress infection. If this treatment failed, antiseptics (octenisept and betadine) were used. Fluoroquinolones (ciprofloxacin, ofloxacin, levofloxacin and moxifloxacin (Vigamox)) are nalidixic acid derivatives and inhibit (1) bacterial DNA-gyrase, a tetramer consisting of two A (GyrA) and two B subunits (GyrB), and (2) topoisomerase IV, a tetramer consisting of two C (ParC) and two E (parE) subunits. These enzymes (DNA gyrase and topoisomerase IV) are involved in DNA replication, recombination and restoration. Fluoroquinolones inhibit these enzymes, thereby impairing DNA reproduction. The chromosomal DNA of the organism becomes degraded, which results in the death of the organism. Second-generation fluoroquinolones (ofloxacin) have low affinity for topoisomerase IV. This fact might be the reason for limited ofloxacin activity against staphylococci and streptococci as well as for fast development of resistance to ofloxacin of these cocci. The primary target for quinolones in Gram-negative bacteria (such as Pseudomonas aeruginosae, Esсherichia coli and Neisseria gonorrheae) is DNA gyrase, whereas in Gram-positive bacteria, such as S. aureus and S. pneumoniae, the primary target is topoisomerase IV. Fluoroquinolones not only kill bacteria, but also inhibit bacterial growth for 2-6 hours after application. Although the bactericidal activity of second-generation fluoroquinolones is comparable to that of levofloxacin, the latter is more active against gram-positive organisms [1, 2]. Floximed eye drops (pH4.5) are poorly dissolved in tear fluid (pH7.4), which results in accumulation of deposits in the stroma, inactivation of the antibiotic, and decreased epithelialization rates. Since levofloxacin eye drops have a close-to-neutral pH6.5, levofloxacin does not leave deposits on the corneal surface as opposed to Floximed. Among the fluoroquinolones that have been studied (levofloxacin, ofloxacin, ciprofloxacin, moxifloxacin, and gatifloxacin), levofloxacin was found to be less cytotoxic to both human corneal endothelial cells and keratocytes [5]. Moxifloxacin is a fourth-generation fluoroquinolone. Compared to levofloxacin, moxifloxacin has demonstrated lower bactericidal activity against Gram-positive microbes, but higher bactericidal activity against Gram-positive pathogens; in addition, it is more toxic. Aminoglycosides (e.g., gentamycin and tobramycin (tobrimed)) received their name because their molecule consists of amino sugars linked through glycosidic bonds to an amynociclitol. They penetrate the bacterial cytoplasmic membrane and inhibit protein synthesis by binding to the 30S subunit of ribosomes. In addition, they are effective against both growing and resting organisms. Aminoglycoside resistance develops as a consequence of (1) the activity of several enzymes that deactivate them (Esсherichia coli, Pseudomonas, Staphylococcus) or (2) decreased bacterial cell permeability (amikacin) due to altered transpost in anaerobes. The bactericidal mode of action of aminoglycosides is due to inhibition of protein synthesis in the bacterial cell and alteration in the permeability of the cytoplasmic membrane. Introduction of second- and third-generation aminoglycosides into clinical practice has been associated with the emergence of bacterial strains with resistance to first-generation aminoglycosides as well as their high activity against Pseudomonas aeruginosa. Aminoglycosides easily penetrate the blood-brain barrier, and, when instilled, they find their way to the stroma, anterior chamber aqueous humor and vitreous. Bacterial resistance to aminoglycosides, when developed, reduces their efficacy against the pathogens. Futaron, an antibacterial agent, is a fusidic acid derivative manufactured by World Medicine Ophthalmics. Fusidic acid is a member of the fusidane class of antimicrobial compounds and has a bactericidal mode of action which is associated with inhibition of protein synthesis in bacterial cells. Fusidanes block the binding of elongation factor G to ribosome with guanosine triphosphate; this interrupts the release of energy required for protein synthesis, thereby leading to bacterial cell death. Futaron (proprietary eye drops) is a 1% suspension of fusidic acid in carbomer 940 gel which is used both to combat microbes and to protect the cornea. Another advantage of this agent is its long-term effect (up to 12 hours) on the contents of the conjunctival sac. Therefore, as opposed to other antibiotics, it is sufficient to instill it twice a day. As a result, the level of antibiotic in the tear fluid increases almost evenly over 12 hours, with antibiotic content becoming high as early as in an hour, and achieving maximum in eight hours. Octenisept (Sch?lke & Mayr GmbH, Norderstedt, Germany), a combination of 0.1% octenidine dihydrochloride and 2% 2-phenoxyethanol, is a colorless and almost odorless aqueous antiseptic solution. It is not absorbed through the mucous membranes, skin or wound surface, and its antimicrobial effect is exhibited through hydrophobic interaction of octenidine dihydrochloride and phenoxyethanol with cytoplasmic membranes of pathogens. Octenisept is not toxic for human tissues, and does not damage tissue cells while eliminating microorganisms. In our study, patients were treated with undiluted (spray) or 1:2 to 1:6 diluted Octenisept by instillation into the conjunctival sac. Patients may report mild burning after instillation of Octenisept; however, this sensation is less pronounced in high dilution of the solution. The medication becomes effective 30 minutes after application and has a long-term efficacy. It must not be mixed with iodine-based preparations. Betadine (EGIS, Budapest, Hungary) is a broad spectrum antiseptic that is bactericidal and effective against some spores and viruses. The antimicrobial effect of polyvinylpyrrolidone (the complex of iodine with polyvinylpyrrolidone), the active component of Betadine, is caused by the oxidative damage and iodine-induced damage to the membrane of the wall cell of the pathogen. Once polyvinylpyrrolidone reaches the cell wall, the free iodine released reacts with cell protein, forming iodamines, and coagulates them; this results in elimination of microorganisms. Povidone-iodine has been widely used in surgical practice for more than 30 years. It exerts immediate bactericidal action against Gram-positive as well as Gram-negative bacteria, with exception of Mycobacterium tuberculosis. Povidone-iodine has been also shown to be effective against fungi, protozoa, and viruses. Since Betadine has a large complex molecule, it does not readily penetrate biological barriers, and the tissue penetration depth is of about 1 mm. Betadine has the additional benefit of releasing its iodine slowly over a period of time, giving it a prolonged bactericidal effect. Patients receive 5% povidone-iodine or 1% povidone-iodine topically. Betadine is a mild topical irritant and is used 2-3 times a day. We investigated the organisms cultured from the conjuinctival samples of ocular burn patients to determine their sensitivity to various antibiotics and antiseptics commonly used in current ophthalmological practice, and to select the most appropriate antibacterial therapy. Five hundred fifty nine cultures were performed, and culture results were analyzed for infecting microbiological agent and antibiotic resistance patterns (Table 2).
Staphylococcus epidermidis, the most commonly cultured organism (52.4%), exhibited various sensitivities to different medications. It showed high sensitivity rates to moxifloxacin, octenisept and futaron (95%, 99.7% and 85.05%, respectively), and low sensitivity rates to ofloxacin and betadine (50% and 40.9%, respectively), while its sensitivity rates to other medications (ciprofloxacin, levofloxacin, gentamycin and tobramycin) were within the range of 71% to 77%. Staphylococcus aureus, the second most common cultured organism (14.3%), was less sensitive to medications under investigation compared to S. epidermis. It showed high sensitivity rates to moxifloxacin and octenisept (99.3% and 76%, respectively) and low sensitivity rates to ofloxacin and betadine (42.8% and 4.9%, respectively), while its sensitivity rates to other medications were within the range of 60.9% to 71.4%. Escherichia coli, the third most common cultured organism (7.6%), was found to be highly sensitive to fluoroquinolones (moxifloxacin, 89%; ciprofloxacin, 78%; levofloxacin, 75%; ofloxacin, 72.5%) and aminoglycosides (tobramycin, 72.5%; and gentamycin, 70.4%) and rather lowly sensitive to Betadine (6.3%) and Futaron (5.7%). Staphylococcus haemolyticus was cultured from 2.5% of the conjunctival samples examined, and demonstrated high sensitivity rates to moxifloxacin and octenisept (100% and 94%, respectively) and low sensitivity rate to Betadine (1.6%), while its sensitivity rates to other medications were within the range of 58.0% to 71.4%. Enterococcus (5.6%) showed the highest sensitivity rates to moxifloxacin and octenisept (99.5% and 78%, respectively) and moderate sensitivity rates to aminoglycosides (gentamycin, 74.5%; and tobramycin, 67.5%) and ciprofloxacin, ofloxacin, and levofloxacin (62%, 52%, and 55%, respectively). However, it was not sensitive to Betadine and Futaron (1% and 0.5%, respectively). As Moraxella, Corynebacterium xerosis and Streptococcus haemolyticus were cultured rarely (4, 9, and 3 positive conjunctival culture specimens, respectively), it was difficult to assess their sensitivity rates to medications under investigation. Pseudomonas aeruginosa was found in 1.5% of the samples obtained, and showed a high sensitivity rate to levofloxacin (82%) and a low sensitivity rate to octenisept (18%). However, it is not sensitive to other antibiotics. Fungi are rather often found in conjunctival samples of ocular burn patients. The incidence of detection of fungi colonies increases with increase in duration of antibacterial treatment of these patients. Fungi cause severe infectious complications such as deep infiltrates, persistent ulcers, and corneas abscesses. Therefore, in a long-term treatment of burn disease, timely prescription of medications for prevention of fungal infection as well as intensive treatment of infectious complications are important. In our ocular burn patients, fluconazole and nizoral were applied either topically and/or systemically, 2% boric acid, betadine, and amphotericinum В were applied topically, and itraconazole was applied systemically. Conclusions First, in the presence of burn process in the eye, conjunctival samples were taken, and various microorganisms that can cause infectious complications were cultured from 88% of these samples. Therefore, following ocular burns, conjunctival specimens should be regularly taken and examined for microorganisms to determine the sensitivity of these organisms to antibacterial medications. Second, administration of antibacterial medications for prevention and treatment of infectious complications should be based on the sensitivity of the organisms to these medications. The sensitivity patterns to medications should be regularly re-examined due to frequent changes both in the composition and sensitivity of conjunctival microflora. Finally, prior to obtaining the results of sensitivity analyses of cultured microorganisms, or if such tests are unavailable, broad spectrum antibiotics, fluoroquinolones (levofloxacin (Levofloximed), ciprofloxacin (Floximed), and moxifloxacin (Vigamox)) and aminoglycosides (gentamycin and tobramycin (Tobrimed)) should be administered. If the microorganism exhibits no sensitivity to antibiotics, such antiseptics as octenisept or betadine may be administered. Our findings demonstrate that these antiseptics have high antibacterial activity against the conjunctival microorganisms cultured from ocular burn patients.
References 1. Puchkovskaya NA, Iakymenko SA, Nepomiaschaia VM. [Ocular burns]. Moscow: Meditsina; 2001 Russian 2. Iakymenko SA, Khrustaliova EA, Molodaia AL. [Features of conjunctival mircroflora of ocular burn patients and its sensitivity to antibacterial agents]. Oftalmol Zh. 2008; 1:37-41 Russian 3. Egorov EA, Astakhov IuS, Stavitskaya TV. [Ocular pharmacology]. Moscow: GEOTAR-Media; 2002 Russian 4. Egorov EA, Alekseev VN, Astakhov IuS. [Rational pharmacotherapy in ophthalmology: A guide for practitioners]. 2nd Edition. Moscow: Literra; 2011 Russian 5. Davis R, Bryson HM. Levofloxacin: A review of its antibacterial activity, pharmacokinetic properties and therapeutic use. Drugs. 1994;47:471-505 6. Klimniuk SL, Sytnyk IO, Tvorko MS, Shyrobokov VP. [Practical microbiology]. Ternopil: Ukrmedknyga, 2004 Ukrainian 7. Pozdeiev OK, Pokrovskii VI. [Medical microbiology]. Moscow: GEOTAR-Meditsina; 1999 Russian 8. Donnenfeld ED. Penetration of topically applied ciprofloxacin, norfloxacin, and ofloxacin into the aqueous humor. Ophthalmology. 1994 May;101(5):902-5
|