J.ophthalmol.(Ukraine).2016;1:19-23.
|
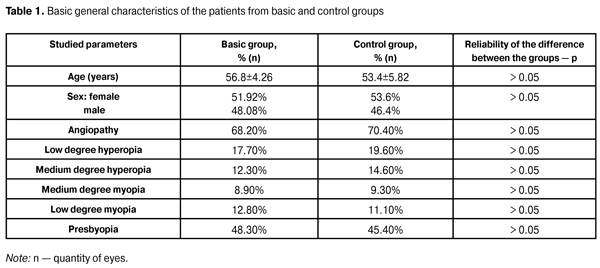
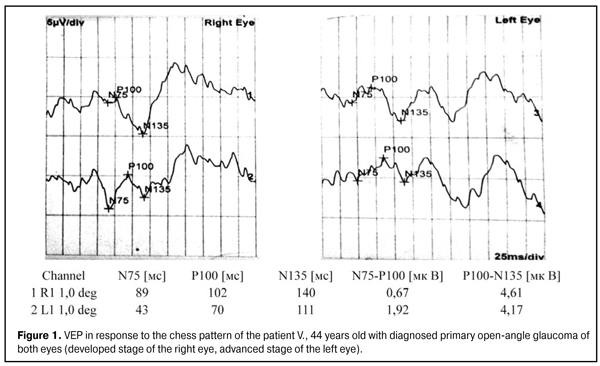
https://doi.org/10.31288/oftalmolzh201611923 Study of visual evoked potentials in clinical practice in the patients with primary open-angle glaucoma L.M. Stotska, Cand. Sc (Med) I.B. Chokova, Cand. Sc (Med) K.S. Romodanova V.B. Reshetniak State Institution “The Filatov Institute of Eye Diseases and Tissue Therapy of the National Academy of Medical Sciences of Ukraine” Odessa (Ukraine) Introduction. It is known that in clinical practice there are no specific neurophysiological tests for diagnosis of glaucoma; however, quite often neurophysiological examination reveals changes which are asymptomatic for a long time before the clinical manifestations, which is especially important for the early diagnosis of glaucoma. Purpose of the study is to make a dynamic analysis of visual evoked potentials in patients with primary open-angle glaucoma at different stages of the disease. Material and Methods: a comprehensive clinical and neurophysiological study of 186 patients (358 eyes) with the diagnosis of primary open-angle glaucoma and in patients whose diagnosis was a subject to confirmation. Results: Based on the analysis of the results of neurophysiological studies, visual evoked potentials in response to pattern and flash, we saw a statistically significant difference between the initial and advanced stages in terms of the latent period P100 (Z = 2.33; p-value = 0.028), the amplitude of peaks P100-N135 (Z = 3.50 p-value = 0.00046), the amplitude of peaks P100-N75 (Z = 2.04; p-value = 0.04); between the advanced and far-advanced stages in terms of the latent period P100 (Z = 3.08; p-value = 0.033). According to the other studied estimates of indicators of visual evoked potentials in response to pattern and flash we did not find any significant differences. Conclusion. Usage of neurophysiological study methods visual evoked potentials in response to pattern is necessary for confirmation of the diagnosis and stage of development of glaucoma during the early diagnosis and the choice of further tactics of treatment. Key words: primary open-angle glaucoma, visual evoked potentials, visual cortex Introduction Glaucoma is a principle cause of irreversible vision loss [5, 6, 15]. However, the question of pathogenesis of glaucoma remains a subject of discussion as for the moment [2]. At the moment, the only proven method of preserving of visual function with glaucoma is a decrease of intraocular pressure (IOP) to the individual safe ("target") level [6]. At the same time, regardless of the level of decrease of IOP after medical treatment, the risk of progression of glaucoma persists. [14]. Studies have shown that in 17-25% of patients a postoperative decrease of IOP does not prevent the progression of glaucomatous optic neuropathy [3, 5, 6]. There are various reasons for this progression. In some studies it has been caused by presence of intracranial pathology in form of local or systemic vascular sclerosis [2, 3, 11], in others ? by decrease in cardiac output which often occurs in elderly patients and with hypertension, and manifests at glaucoma in form of decrease of regional velocity and volume parameters of the blood flow [2, 3]. Some studies have pointed to the decrease of volumetric blood flow in glaucoma operated patients associating it with violation of vascular autoregulation of the disc of optic nerve [3, 9]. The investigations of structural and neurophysiological processes in higher parts of the visual analyzer in patients with primary glaucoma help in the study of primary damage of the structures at this pathology. A number of studies have indicated the defects in structures of the central parts of the brain in patients with primary glaucoma: in the optic nerve there has been observed macroscopically a severe atrophy with loss of significant number of axons, macroscopically lateral geniculate body has been less in size than normal, which may indicate the loss of significant number of neurons due to degenerative changes in it. Microscopic study has found reducing of the radius of neurons and their nuclei, grumose, granular cytoplasm, and a large amount of lipofuscin pigment ? one of the markers of atrophy. In the visual cortex of the patient with primary glaucoma there has been visually detected a decrease of the thickness of cell layer. Degenerative changes in visual cortex at the level of calacarine fissure have been detected most clearly [1, 8]. R. Frena and G. Gainotti have examined the patients with right-sided and left-sided brain lesions and compared them with the patients with the undamaged brain, used as a control group. The authors have performed the tests to study abilities of the patients in visual space perception (VSP) and graphomotor actions in each group separately. This has showed that violations in visual perception or visual-motor activity can result from any damage of hemisphere cortex [4]. In his article G. Gainotti has cited the results of research, which enable him to say that the patients with right-sided and left-sided lesions of cortex have similarities in the violation of visual space perception, but violations of the visual-motor perception and violations of perception of relevant characters are more common in patients with injuries of the left hemisphere, and a unilateral violation of the visual space perception - in patients with right-side injuries [4]. G. Gainotti and colleagues have studied the role of hemispheric asymmetry in the process of visual spatial memorization in patients with injuries of one hemisphere. The results of the correct choice have been better in the control group than in the group of patients with brain injuries, and the patients with right-sided lesions made the right choice more often, than the patients with left-sided lesions. [4] According to structural asymmetry, the functional asymmetry has been identified that characterizes the neurophysiological processes in brain hemispheres. Generally, the main centers responsible for conversational activity are located in the left hemisphere, while the right hemisphere is responsible for processing of information obtained through visual space perception. These findings are confirmed by the studies of relevant violations of visual space perception and visual motor function in patients after stroke [4]. The most important information about processes in the visual cortex has been obtained from the research of evoked potentials of the brain, including the most significant - visual evoked potentials (VEP). It has been reported about the effectiveness of multifocal VEP (mfVEP) for objective detection of the lesion of optic nerve with glaucoma, ischemic optic neuropathy and unilateral optic neuritis. Some defects of the visual field have not been detected because of the weak responses on the better eye, during the comparative evaluation of mfVED from both eyes there has been a correlation of the results of obtained mfVEP with the results of computed Humphrey-perimetry [7, 11]. There is an opinion that VEP changes in response to flash are not specific for any lesion of the visual system. Thus, increase of VEP latency components can result from the damaged transmission of impulses in the retina, demyelination of nerve fibers at different levels of optic tract - from the optic nerve to the cortex of occipital lobe or synaptic abnormalities in the retina or the visual cortex [7]. We know that in clinical practice, there are no specific neurophysiological tests for diagnosis of glaucoma, but neurophysiological examination often detects changes which occur asymptomatic for a long time till the clinical manifestations, which is especially important for early diagnosis of glaucomatous process [7]. The purpose of the study is to make a dynamic analysis of visual evoked potentials in clinical practice in patients with primary open-angle glaucoma at different stages of glaucoma process. Materials and Methods On the basis of the State Institution “The Filatov Institute of Eye Diseases and Tissue Therapy of the National Academy of Medical Sciences of Ukraine”, we conducted a comprehensive clinical and neurophysiological study of 186 patients (358 eyes) with diagnosed primary open-angle glaucoma (POAG) or those patients whose diagnosis was subject to confirmation. Among them there were 81 women (51.92%) and 75 men (48.08%), average age was (56.8 ± 4.26) years at different stages of glaucoma process (study group). All patients underwent clinical research methods: visometry, tonometry, tonography, refractometry, biomicroscopy, direct and inverse ophthalmoscopy, computed perimetry on the device "Humphrey" ("ZEISS, USA)", determining of lability and sensitivity threshold of optic nerves for phosphene, optical coherent tomography in the device "OPTOPOL" (SOCT Copernicus, Poland). For the diagnosis of pathological conditions, neurophysiological research methods were also used, i.e. visual evoked potentials (VEP) in response to chess pattern and flash. During registration of latency and amplitude of peaks in all leads the intraocular difference of these data was analyzed. VEP studies were performed using the device "RETIscan" (Roland Sonsult, Germany). To characterize the pathological process, changes in the duration of latent period of VEP in response to chess pattern, changes in latent period of the main peaks N75, P100, N135, changes in the amplitude N75-P100, P100-N75; VEP in response to flash - changes of latent period of the main peaks N1, P1, and amplitude N1-P1 were considered. The above parameters are important to assess both VEP dynamics and degree of increase of pathological process. The results of the basic group (158 persons; 298 eyes) were analyzed depending on the stage of POAG and were respectively consisted of four subgroups: subgroup I: preglaucoma (42 patients; 84 eyes); subgroup II: early stage (48 patients; 96 eyes); subgroup III: developed stage (36 patients; 65 eyes); and subgroup IV: advanced stage (30 patients; 53 eyes). Subgroup of preglaucoma (suspected glaucoma) was formed from the patients whose results differed from the standard, according to the above studies in one or two indicators (reoophtalmography, OCT, tonometry, tonography ? intraocular difference or daily tonometry more than 3-4 mm). Patients with the end-stage POAG and high-stage ametropia were excluded from the study. The control group included 30 patients without a diagnosis of POAG, with slight or moderate emmetropia, and was comparable to the main group by age, sex and other diseases (Table. 1).
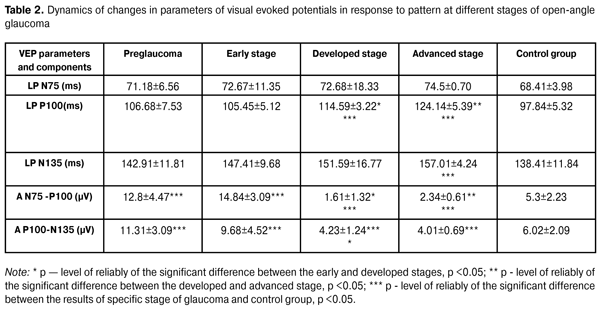
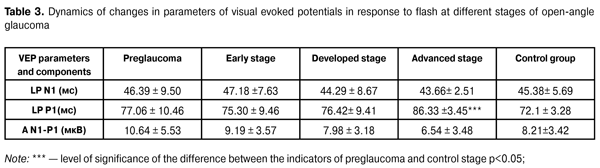
No patients of the basic and control group had any optic nerve disease, macular dystrophy, hypertensive retinopathy, blurred optical media, diabetes, craniocerebral trauma, acute and chronic disorders of cerebral circulation. The data of visual evoked potentials except those of control group was compared with data from the literature [15] and also data about the standards of specific laboratory where researches were performed as there were no recognized data standards in clinical practice. Statistical data processing was performed using the package Statistica 10 using mean values of standard deviation, reliability of the results of difference ? p (paired test with two-sided distribution) less than 0.05, and using non-parametric Mann-Whitney method, criteria analysis chi-square, Spirmann criterion. Results All patients, 156 persons (298 eyes) of the basic group and 30 persons (60 eyes) of the control group, underwent a full range of research of VEP in response to pattern and flash, in left and right eyes separately. Table 1 shows that no statistically significant differences between the basic and control group were found according to such indicators as age, sex and other physical illness. Using a non-parametric method with the criterion chi-square, we analyzed the patients’ data at different stages of glaucoma process. According to the data obtained in patients with suspected glaucoma according to the indicators N75 and P100, the duration of latent period in 96.1% (p <0.05) and 86.2% of eyes (p <0.05) was within the normal range, respectively; N75-P100 and P100-N135 peak amplitude within the normal range was observed in 21.4% (p <0.05) and 34.5% (p <0.05) of patients, respectively. Increase of these amplitudes above the standard (according to the data the standards for equipment and laboratories in which the research was performed, were noted in 78.6% (p <0.05) and 65.5% (p <0.05) of eyes respectively - so-called "supernormal" results. 86.4% (p <0.05) and 81.2% (p <0.05) of patients with early stage had the latent period for indicators N75 and P100 within the normal range, respectively; N75- P100 and P100-N135 peak amplitude was within the normal range in 34.85% (p <0.05) and 41.86% (p <0.05) respectively, increase of the above amplitudes was observed in 65.15% (p < 0.05) and 58.14% (p <0.05), respectively. Pathological extension of the latent period according to indicators N75 and P100 was observed in 12.31% (p> 0.05) and 74.55% (p <0.05) of patients with advanced stage, respectively; decrease of N75-P100 peak amplitude with change of P100 peak top in W-shaped form, indicating the gross violation of the optic nerve conduction observed in 86.42% (p <0.05) of cases; decrease of R100- N135 peak amplitude was observed in 64.51% (p <0.05). Patients with the advanced stage showed significant extension of latent period P100 in 84.61% (p <0.05) of patients and decrease of N75-P100 peak amplitude with change of P100 peak top in W-shaped form in 79.14% (p <0 , 05) of patients; decrease of R100- N135 peak amplitude in 68.43% (p <0.05) (Table 2).
In terms of Mann-Whitney criterion, we observed a statistically reliable difference between the early and developed stages according to the indicators of P100 latent period (Z = 2.33; p = 0.028), P100-N135 peak amplitude (Z = 3.50, p = 0.00046), N75-P100 peak amplitude (Z = 2.04; p = 0.04); between the developed and advanced stages according to the indicator of P100 latent period (Z = 3.08; p = 0.033). According to other investigated indicators of visual evoked potentials in response to flash and pattern according to Mann-Whitney criterion, no reliable statistical differences were found.
Patients with suspected glaucoma during the second examination after 6 months and 1 year confirmed the initial diagnosis of primary open-angle glaucoma according to the general clinical research in 78.4% of cases. When used the additional neurophysiological methods of examination found more pronounced changes of pathological process in 34.80% of patients with early stage and they were transferred to the group - developed stage of glaucoma, with appropriate changes in the tactics of further treatment. Similar changes were made in the group with developed glaucoma - 14% of patients were transferred to the group of advanced stage of glaucoma.
Conclusions Considering the dynamics of changes in the parameters of VEP in response to the chess pattern and flash in patients at different stages of primary open-angle glaucoma, we observe the following: 1. Bioelectric activity of the cerebral cortex, according to the visual evoked potential in response to pattern in patients with suspected glaucoma and early stage during analysis of duration of the latent period respectively in 96.1% and 86.36% of cases statistically significant showed "supernormal" results. And only the patients with developed stage (74.55%) and patients with the advanced stage (84.61% of cases) showed a statistically significant extension of the latent period. 2. Amplitude of fluctuation of the main peaks N75-P100 was significantly higher than normal in 78.61% and 65.15% cases respectively in patients with suspected glaucoma and early stage of primary open-angle glaucoma; patients with developed and advanced stages of glaucoma showed a statistically reliable decrease of the amplitude of fluctuation peaks and change in P100 top peak in W-shaped form. 3. Usage of neurophysiological research methods - visual evoked potentials in response to pattern – is important for confirmation of diagnosis and stage of development of glaucoma for early diagnosis and the choice of further tactics of treatment. References 1. Alekseev VI, Nikitin DI, Gazizova IR. [Changes in the central parts of visual analyzer in case of primary open-angle glaucoma]. Collection of abstracts from research and practice conference “Filatov Readings – 2012”. Odessa. 2012: 95. Russian. 2. Zavgorodnyaya NV, Pasechnikova NV. [Primary glaucoma. Fresh approach to the old problem]. Zaporozhzhye. 2010:192. Russian. 3. Kisseleva OA, Makuha SA, Yakubova AV, Vasilenkova LV. [Examination of eye haemodynamics in patients with primary open angle glaucoma after fistulizing surgery]. Rossiysky oftalmologicheskiy zhurnal. 2014;7(2):94-96. Russian. 4. Lessella S, Van Dalena JTU. [Neuroophthalmology]. Moscow: Meditsina. 1983:99-101. Russian. 5. Moiseenko RA, Golubchikov MV, Slabkiy GO, Rykov SA. et al. [Ophthalmological care in Ukraine in 2006-2011]. Kiev;2:183. Russian. 6. Rykov SA, Vitovskaya OP, Shargorodskaya IV et al. [Unified clinical protocol of medical care. Primary open angle glaucoma. Primary and secondary care]. Order of the Ministry of Health of Ukraine No. 816, dated 24.11.2011. Kiev. 2011:37. Russian. 7. Shamshinova АМ , Andreeva TM. [Clinical physiology of sense of vision]. Moscow. 2006:956. Russian. 8. Alekseev V, Gаzizova I. Meaning of neurodegeneration in the pathogenesis of glaucoma, 11 th EGS Congress. Nice. 2014:60. 9. Berisha F et al. Effect of trabecullctomy on ocular blood flow. Br. J. Ophthalmol. 2005;89 (2):185-8. 10. Hasegawa S, Abe H. Mapping of glaucomatous visual field defects by multifocal VEPs. Inv. Ophthalmol. Vis.Sci. 2001;42:3341–3348. 11. Hood DC, Odel JG, Winn BJ. The multifocal visual evoked potential. J. Neuroophthalmol. 2003;23(4):279-289. 12. Klistorner A, Graham SL. Objective perimetry in glaucoma. Ophthalmol. 2000;107:2283–2299. 13. Klistorner A, Graham SL, Grigg JR. et al. Multifocal topographic visual evoked potential: improving objective detection of local visual field defects. Inv. Ophthalmol. Vis. Sci. 1998;39(6):937 – 950 14. Nesher R, Mimouni M, Epshtein E, Gorck L, Trick C. Reported status OCT retinal nerve fiber Layer thicrness measurements. In: Natsionaln. zhurn. glaukomy. 2014;2(13):11-15. 15. Quigley UA. Glaucoma. Lancet. 2011:16;377(9774):1367–7. 16. Wu De Zheng, Liu Yan Atlas of testind and clinical application for Roland Electrophysiolodikal Instrument. Beigind science and technology Press. China, 2006:5-19.
|